Small RNA Sequencing Reveals a Distinct MicroRNA Signature between Glucocorticoid Responder and Glucocorticoid Non-Responder Primary Human Trabecular Meshwork Cells after Dexamethasone Treatment
- PMID: 38002955
- PMCID: PMC10671261
- DOI: 10.3390/genes14112012
Small RNA Sequencing Reveals a Distinct MicroRNA Signature between Glucocorticoid Responder and Glucocorticoid Non-Responder Primary Human Trabecular Meshwork Cells after Dexamethasone Treatment
Abstract
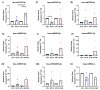
Glucocorticoids (GCs) are known to regulate several physiological processes and are the mainstay in the management of inflammatory eye diseases. The long-term use of GC causes raised intraocular pressure (IOP) or ocular hypertension (OHT) in about 30-50% of the susceptible individuals depending on the route of administration, and can lead to steroid-induced secondary glaucoma. The present study aims to understand the role of microRNAs (miRNAs) in differential glucocorticoid (GC) responsiveness in human trabecular meshwork (HTM) cells using small RNA sequencing. The human organ-cultured anterior segment (HOCAS) model was used to identify whether donor eyes were from GC-responders (GC-R; n = 4) or GC-non-responders (GC-NR; n = 4) following treatment with either 100 nM dexamethasone (DEX) or ethanol (ETH) for 7 days. The total RNA was extracted from cultured HTM cells with known GC responsiveness, and the differentially expressed miRNAs (DEMIRs) were compared among the following five groups: Group #1: ETH vs. DEX-treated GC-R; #2: ETH vs. DEX-treated GC-NR; #3: overlapping DEGs between Group #1 and #2; #4: Unique DEMIRs of GC-R; #5: Unique DEMIRs of GC-NR; and validated by RT-qPCR. There were 13 and 21 DEMIRs identified in Group #1 and Group #2, respectively. Seven miRNAs were common miRNAs dysregulated in both GC-R and GC-NR (Group #3). This analysis allowed the identification of DEMIRs that were unique to GC-R (6 miRNAs) and GC-NR (14 miRNAs) HTM cells, respectively. Ingenuity Pathway Analysis identified enriched pathways and biological processes associated with differential GC responsiveness in HTM cells. This is the first study to reveal a unique miRNA signature between GC-R and GC-NR HTM cells, which raises the possibility of developing new molecular targets for the management of steroid-OHT/glaucoma.
Keywords: glaucoma; glucocorticoids; intra-ocular pressure; miRNA; microRNAs; ocular hypertension; pathway analysis; small RNA-Seq; trabecular meshwork.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






Similar articles
-
A Comparative Genome-Wide Transcriptome Analysis of Glucocorticoid Responder and Non-Responder Primary Human Trabecular Meshwork Cells.Genes (Basel). 2022 May 15;13(5):882. doi: 10.3390/genes13050882. Genes (Basel). 2022. PMID: 35627267 Free PMC article.
-
Hsa-MiR-483 -3p regulates the extracellular matrix proteins via TGFβ2/SMAD4 signaling in the glucocorticoid-responsive human trabecular meshwork cells.Cell Tissue Res. 2025 Aug;401(2):145-154. doi: 10.1007/s00441-025-03985-z. Epub 2025 Jun 5. Cell Tissue Res. 2025. PMID: 40471287
-
A Comparison of Gene Expression Profiles between Glucocorticoid Responder and Non-Responder Bovine Trabecular Meshwork Cells Using RNA Sequencing.PLoS One. 2017 Jan 9;12(1):e0169671. doi: 10.1371/journal.pone.0169671. eCollection 2017. PLoS One. 2017. PMID: 28068412 Free PMC article.
-
Role of the alternatively spliced glucocorticoid receptor isoform GRβ in steroid responsiveness and glaucoma.J Ocul Pharmacol Ther. 2014 Mar-Apr;30(2-3):121-7. doi: 10.1089/jop.2013.0239. Epub 2014 Feb 7. J Ocul Pharmacol Ther. 2014. PMID: 24506296 Free PMC article. Review.
-
Effects of glucocorticoids on the trabecular meshwork: towards a better understanding of glaucoma.Prog Retin Eye Res. 1999 Sep;18(5):629-67. doi: 10.1016/s1350-9462(98)00035-4. Prog Retin Eye Res. 1999. PMID: 10438153 Review.
Cited by
-
Influence of dexamethasone-induced matrices on the TM transcriptome.Exp Eye Res. 2024 Nov;248:110069. doi: 10.1016/j.exer.2024.110069. Epub 2024 Sep 2. Exp Eye Res. 2024. PMID: 39233306
-
Integrated Bioinformatics-Based Identification and Validation of Neuroinflammation-Related Hub Genes in Primary Open-Angle Glaucoma.Int J Mol Sci. 2024 Jul 26;25(15):8193. doi: 10.3390/ijms25158193. Int J Mol Sci. 2024. PMID: 39125762 Free PMC article.
References
-
- Gordon D.M. Hormonal-steroid therapy in ocular inflammations. Seminar. 1955;17:11–14. - PubMed
-
- Gregori N.Z., Rosenfeld P.J., Puliafito C.A., Flynn H.W., Lee J.E., Mavrofrides E.C., Smiddy W.E., Murray T.G., Berrocal A.M., Scott I.U., et al. One-year safety and efficacy of intravitreal triamcinolone acetonide for the management of macular edema secondary to central retinal vein occlusion. Retina. 2006;26:889–895. doi: 10.1097/01.iae.0000237111.82357.30. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

