iTRAQ-Based Phosphoproteomic Analysis Exposes Molecular Changes in the Small Intestinal Epithelia of Cats after Toxoplasma gondii Infection
- PMID: 38003154
- PMCID: PMC10668779
- DOI: 10.3390/ani13223537
iTRAQ-Based Phosphoproteomic Analysis Exposes Molecular Changes in the Small Intestinal Epithelia of Cats after Toxoplasma gondii Infection
Abstract
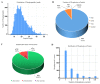
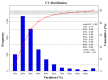
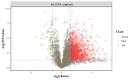
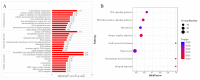
Toxoplasma gondii, an obligate intracellular parasite, has the ability to invade and proliferate within most nucleated cells. The invasion and destruction of host cells by T. gondii lead to significant changes in the cellular signal transduction network. One important post-translational modification (PTM) of proteins is phosphorylation/dephosphorylation, which plays a crucial role in cell signal transmission. In this study, we aimed to investigate how T. gondii regulates signal transduction in definitive host cells. We employed titanium dioxide (TiO2) affinity chromatography to enrich phosphopeptides in the small intestinal epithelia of cats at 10 days post-infection with the T. gondii Prugniuad (Pru) strain and quantified them using iTRAQ technology. A total of 4998 phosphopeptides, 3497 phosphorylation sites, and 1805 phosphoproteins were identified. Among the 705 differentially expressed phosphoproteins (DEPs), 68 were down-regulated and 637 were up-regulated. The bioinformatics analysis revealed that the DE phosphoproteins were involved in various cellular processes, including actin cytoskeleton reorganization, cell necroptosis, and MHC immune processes. Our findings confirm that T. gondii infection leads to extensive changes in the phosphorylation of proteins in the cat intestinal epithelial cells. The results of this study provide a theoretical foundation for understanding the interaction between T. gondii and its definitive host.
Keywords: Toxoplasma gondii; cat; phosphoproteomics; post-translational modification.
Conflict of interest statement
The authors declare that they have no competing interest.
Figures



Similar articles
-
iTRAQ-Based Global Phosphoproteomics Reveals Novel Molecular Differences Between Toxoplasma gondii Strains of Different Genotypes.Front Cell Infect Microbiol. 2019 Aug 23;9:307. doi: 10.3389/fcimb.2019.00307. eCollection 2019. Front Cell Infect Microbiol. 2019. PMID: 31508380 Free PMC article.
-
Global proteomic profiling of multiple organs of cats (Felis catus) and proteome-transcriptome correlation during acute Toxoplasma gondii infection.Infect Dis Poverty. 2022 Sep 14;11(1):96. doi: 10.1186/s40249-022-01022-7. Infect Dis Poverty. 2022. PMID: 36104766 Free PMC article.
-
Dynamic RNA profiles in the small intestinal epithelia of cats after Toxoplasma gondii infection.Infect Dis Poverty. 2023 Jul 25;12(1):68. doi: 10.1186/s40249-023-01121-z. Infect Dis Poverty. 2023. PMID: 37491273 Free PMC article.
-
iTRAQ-based phosphoproteomic analysis reveals host cell's specific responses to Toxoplasma gondii at the phases of invasion and prior to egress.Biochim Biophys Acta Proteins Proteom. 2019 Mar;1867(3):202-212. doi: 10.1016/j.bbapap.2018.12.004. Epub 2018 Dec 18. Biochim Biophys Acta Proteins Proteom. 2019. PMID: 30576742
-
[The impact of Toxoplasma gondii infection on host cell signal transduction].Zhongguo Ji Sheng Chong Xue Yu Ji Sheng Chong Bing Za Zhi. 2011 Oct;29(5):378-84. Zhongguo Ji Sheng Chong Xue Yu Ji Sheng Chong Bing Za Zhi. 2011. PMID: 24830203 Review. Chinese.
Cited by
-
Incremental Modification in the Existing Approaches for Affinity Chromatographic Enrichment of Phosphoproteins Improves Their Profile in Liquid Chromatography-Tandem Mass Spectrometry Analysis.Anal Sci Adv. 2024 Dec 20;6(1):e202400058. doi: 10.1002/ansa.202400058. eCollection 2025 Jun. Anal Sci Adv. 2024. PMID: 39902464 Free PMC article.
-
Phosphoproteomic profiling of feline mammary carcinoma: Insights into tumor grading and potential therapeutic targets.PLoS One. 2025 Aug 21;20(8):e0330520. doi: 10.1371/journal.pone.0330520. eCollection 2025. PLoS One. 2025. PMID: 40839607 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

