Evaluating Manganese, Zinc, and Copper Metal Toxicity on SH-SY5Y Cells in Establishing an Idiopathic Parkinson's Disease Model
- PMID: 38003318
- PMCID: PMC10671677
- DOI: 10.3390/ijms242216129
Evaluating Manganese, Zinc, and Copper Metal Toxicity on SH-SY5Y Cells in Establishing an Idiopathic Parkinson's Disease Model
Abstract
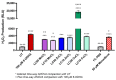
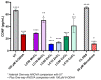
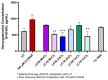
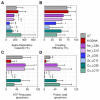
Parkinson's disease (PD) is a neurodegenerative condition marked by loss of motor coordination and cognitive impairment. According to global estimates, the worldwide prevalence of PD will likely exceed 12 million cases by 2040. PD is primarily associated with genetic factors, while clinically, cases are attributed to idiopathic factors such as environmental or occupational exposure. The heavy metals linked to PD and other neurodegenerative disorders include copper, manganese, and zinc. Chronic exposure to metals induces elevated oxidative stress and disrupts homeostasis, resulting in neuronal death. These metals are suggested to induce idiopathic PD in the literature. This study measures the effects of lethal concentration at 10% cell death (LC10) and lethal concentration at 50% cell death (LC50) concentrations of copper, manganese, and zinc chlorides on SH-SY5Y cells via markers for dopamine, reactive oxygen species (ROS) generation, DNA damage, and mitochondrial dysfunction after a 24 h exposure. These measurements were compared to a known neurotoxin to induce PD, 100 µM 6-hydroxydopamine (6-ODHA). Between the three metal chlorides, zinc was statistically different in all parameters from all other treatments and induced significant dopaminergic loss, DNA damage, and mitochondrial dysfunction. The LC50 of manganese and copper had the most similar response to 6-ODHA in all parameters, while LC10 of manganese and copper responded most like untreated cells. This study suggests that these metal chlorides respond differently from 6-ODHA and each other, suggesting that idiopathic PD utilizes a different mechanism from the classic PD model.
Keywords: SH-SY5Y; dose–response curves; hydroxydopamine; neurodegeneration; occupational exposure.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

