IK Channel-Independent Effects of Clotrimazole and Senicapoc on Cancer Cells Viability and Migration
- PMID: 38003471
- PMCID: PMC10671816
- DOI: 10.3390/ijms242216285
IK Channel-Independent Effects of Clotrimazole and Senicapoc on Cancer Cells Viability and Migration
Abstract
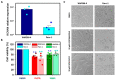
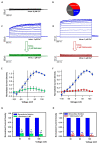
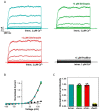
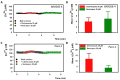
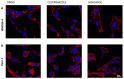
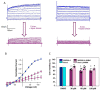
Many studies highlighted the importance of the IK channel for the proliferation and the migration of different types of cancer cells, showing how IK blockers could slow down cancer growth. Based on these data, we wanted to characterize the effects of IK blockers on melanoma metastatic cells and to understand if such effects were exclusively IK-dependent. For this purpose, we employed two different blockers, namely clotrimazole and senicapoc, and two cell lines: metastatic melanoma WM266-4 and pancreatic cancer Panc-1, which is reported to have little or no IK expression. Clotrimazole and senicapoc induced a decrease in viability and the migration of both WM266-4 and Panc-1 cells irrespective of IK expression levels. Patch-clamp experiments on WM266-4 cells revealed Ca2+-dependent, IK-like, clotrimazole- and senicapoc-sensitive currents, which could not be detected in Panc-1 cells. Neither clotrimazole nor senicapoc altered the intracellular Ca2+ concentration. These results suggest that the effects of IK blockers on cancer cells are not strictly dependent on a robust presence of the channel in the plasma membrane, but they might be due to off-target effects on other cellular targets or to the blockade of IK channels localized in intracellular organelles.
Keywords: IK; KCNN4; KCa3.1; blockers; cancer; clotrimazole; melanoma; pancreatic duct adenocarcinoma (PDAC); senicapoc.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
KCa3.1 (IK) modulates pancreatic cancer cell migration, invasion and proliferation: anomalous effects on TRAM-34.Pflugers Arch. 2016 Nov;468(11-12):1865-1875. doi: 10.1007/s00424-016-1891-9. Epub 2016 Oct 17. Pflugers Arch. 2016. PMID: 27752766
-
Blockage of intermediate-conductance Ca2+-activated K+ channels inhibit human pancreatic cancer cell growth in vitro.Mol Pharmacol. 2004 Mar;65(3):630-8. doi: 10.1124/mol.65.3.630. Mol Pharmacol. 2004. PMID: 14978241
-
Inhibition of T cell proliferation by selective block of Ca(2+)-activated K(+) channels.Proc Natl Acad Sci U S A. 1999 Sep 14;96(19):10917-21. doi: 10.1073/pnas.96.19.10917. Proc Natl Acad Sci U S A. 1999. PMID: 10485926 Free PMC article.
-
The Ca2+-activated K+ channel of intermediate conductance:a possible target for immune suppression.Expert Opin Ther Targets. 2002 Dec;6(6):623-36. doi: 10.1517/14728222.6.6.623. Expert Opin Ther Targets. 2002. PMID: 12472376 Review.
-
Targeting Kca3.1 Channels in Cancer.Cell Physiol Biochem. 2021 May 28;55(S3):131-144. doi: 10.33594/000000374. Cell Physiol Biochem. 2021. PMID: 34043300 Review.
Cited by
-
Clotrimazole inhibits growth of multiple myeloma cells in vitro via G0/G1 arrest and mitochondrial apoptosis.Sci Rep. 2024 Jul 4;14(1):15406. doi: 10.1038/s41598-024-66367-5. Sci Rep. 2024. PMID: 38965397 Free PMC article.
-
zDHHC3-mediated S-palmitoylation of SLC9A2 regulates apoptosis in kidney clear cell carcinoma.J Cancer Res Clin Oncol. 2024 Apr 15;150(4):194. doi: 10.1007/s00432-024-05737-y. J Cancer Res Clin Oncol. 2024. PMID: 38619631 Free PMC article.
-
Emerging Therapeutic Options in Pancreatic Cancer Management.Int J Mol Sci. 2024 Feb 5;25(3):1929. doi: 10.3390/ijms25031929. Int J Mol Sci. 2024. PMID: 38339207 Free PMC article.
-
ACYP2 functions as an innovative nano-therapeutic target to impede the progression of hepatocellular carcinoma by inhibiting the activity of TERT and the KCNN4/ERK pathway.J Nanobiotechnology. 2024 Sep 12;22(1):557. doi: 10.1186/s12951-024-02827-4. J Nanobiotechnology. 2024. PMID: 39267048 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

