Production of Proinflammatory Cytokines by CD4+ and CD8+ T Cells in Response to Mycobacterial Antigens among Children and Adults with Tuberculosis
- PMID: 38003817
- PMCID: PMC10675744
- DOI: 10.3390/pathogens12111353
Production of Proinflammatory Cytokines by CD4+ and CD8+ T Cells in Response to Mycobacterial Antigens among Children and Adults with Tuberculosis
Abstract
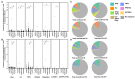
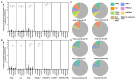
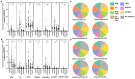
Tuberculosis (TB), caused by Mycobacterium tuberculosis (Mtb), remains a leading cause of pediatric morbidity and mortality. Young children are at high risk of TB following Mtb exposure, and this vulnerability is secondary to insufficient host immunity during early life. Our primary objective was to compare CD4+ and CD8+ T-cell production of proinflammatory cytokines IFN-gamma, IL-2, and TNF-alpha in response to six mycobacterial antigens and superantigen staphylococcal enterotoxin B (SEB) between Ugandan adults with confirmed TB (n = 41) and young Ugandan children with confirmed (n = 12) and unconfirmed TB (n = 41), as well as non-TB lower respiratory tract infection (n = 39). Flow cytometry was utilized to identify and quantify CD4+ and CD8+ T-cell cytokine production in response to each mycobacterial antigen and SEB. We found that the frequency of CD4+ and CD8+ T-cell production of cytokines in response to SEB was reduced in all pediatric cohorts when compared to adults. However, T-cell responses to Mtb-specific antigens ESAT6 and CFP10 were equivalent between children and adults with confirmed TB. In contrast, cytokine production in response to ESAT6 and CFP10 was limited in children with unconfirmed TB and absent in children with non-TB lower respiratory tract infection. Of the five additional mycobacterial antigens tested, PE3 and PPE15 were broadly recognized regardless of TB disease classification and age. Children with confirmed TB exhibited robust proinflammatory CD4+ and CD8+ T-cell responses to Mtb-specific antigens prior to the initiation of TB treatment. Our findings suggest that adaptive proinflammatory immune responses to Mtb, characterized by T-cell production of IFN-gamma, IL-2, and TNF-alpha, are not impaired during early life.
Keywords: Mycobacterium tuberculosis; T cells; adaptive immunity; cytokines; pediatric.
Conflict of interest statement
OHSU and DA Lewinsohn, DM Lewinsohn, and G. Swarbrick have a financial interest in ViTi, a company that may have a commercial interest in the results of this research and technology. This potential conflict of interest has been reviewed and managed by OHSU.
Figures


Similar articles
-
Functional Signatures of Human CD4 and CD8 T Cell Responses to Mycobacterium tuberculosis.Front Immunol. 2014 Apr 22;5:180. doi: 10.3389/fimmu.2014.00180. eCollection 2014. Front Immunol. 2014. PMID: 24795723 Free PMC article. Review.
-
Cell-Mediated Immune Responses to in vivo-Expressed and Stage-Specific Mycobacterium tuberculosis Antigens in Latent and Active Tuberculosis Across Different Age Groups.Front Immunol. 2020 Feb 11;11:103. doi: 10.3389/fimmu.2020.00103. eCollection 2020. Front Immunol. 2020. PMID: 32117257 Free PMC article.
-
Impact of antiretroviral and tuberculosis therapies on CD4+ and CD8+ HIV/M. tuberculosis-specific T-cell in co-infected subjects.Immunol Lett. 2018 Jun;198:33-43. doi: 10.1016/j.imlet.2018.04.001. Epub 2018 Apr 7. Immunol Lett. 2018. PMID: 29635002
-
Clonal analysis of the T-cell response to in vivo expressed Mycobacterium tuberculosis protein Rv2034, using a CD154 expression based T-cell cloning method.PLoS One. 2014 Jun 6;9(6):e99203. doi: 10.1371/journal.pone.0099203. eCollection 2014. PLoS One. 2014. PMID: 24905579 Free PMC article.
-
Mycobacterial Heat Shock Proteins in Sarcoidosis and Tuberculosis.Int J Mol Sci. 2023 Mar 7;24(6):5084. doi: 10.3390/ijms24065084. Int J Mol Sci. 2023. PMID: 36982159 Free PMC article. Review.
References
-
- Barr L. Tuberculosis Research Funding Trends, 2005–2018. 2019. Tuberculosis Research Funding Trends. [(accessed on 1 September 2023)]. Available online: https://www.treatmentactiongroup.org/wp-content/uploads/2019/12/tbrd_201....
-
- World Health Organization Global Tuberculosis Report 2020. 2020. Global Tuberculosis Report. 15 October 2020. [(accessed on 1 September 2023)]. Available online: https://www.who.int/publications/i/item/9789240013131.
-
- Marais B.J., Gie R.P., Schaaf H.S., Hesseling A.C., Obihara C.C., Starke J.J., Enarson D.A., Donald P.R., Beyers N. The natural history of childhood intra-thoracic tuberculosis: A critical review of literature from the pre-chemotherapy era. Int. J. Tuberc. Lung Dis. 2004;8:392–402. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

