Food Administration and Not Genetic Variants Causes Pharmacokinetic Variability of Tadalafil and Finasteride
- PMID: 38003881
- PMCID: PMC10672114
- DOI: 10.3390/jpm13111566
Food Administration and Not Genetic Variants Causes Pharmacokinetic Variability of Tadalafil and Finasteride
Abstract
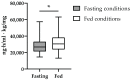
Tadalafil and finasteride are used in combination for the management of benign prostatic hyperplasia (BPH). Genetic variations in genes involved in the metabolism and transport of tadalafil or finasteride (i.e., pharmacogenes) could affect their pharmacokinetic processes altering their drug exposure, efficacy, and toxicity. The main objective of this study was to investigate the effects of variants in pharmacogenes on the pharmacokinetics of tadalafil and finasteride. An exploratory candidate gene study involving 120 variants in 33 genes was performed with 66 male healthy volunteers from two bioequivalence clinical trials after administration of tadalafil/finasteride 5 mg/5 mg under fed or fasting conditions. Afterwards, a confirmatory study was conducted with 189 male and female volunteers receiving tadalafil 20 mg formulations in seven additional bioequivalence clinical trials. Regarding tadalafil, fed volunteers showed higher area in the time-concentration curve (AUC∞), maximum plasma concentration (Cmax), and time to reach Cmax (tmax) compared to fasting volunteers; male volunteers also showed higher AUC∞ and Cmax compared to female volunteers. Furthermore, fed volunteers presented higher finasteride AUC∞, Cmax and tmax compared to fasting individuals. Variants in ABCC3, CYP1A2, CES1, NUDT15, SLC22A1/A2 and UGT2B10 were nominally associated with pharmacokinetic variation in tadalafil and/or finasteride but did not remain significant after correction for multiple comparisons. Genetic variation did not demonstrate to clinically impact on the pharmacokinetics of finasteride and tadalafil; however, additional studies with larger sample sizes are needed to assess the effect of rare variants, such as CYP3A4*20 or *22, on tadalafil and finasteride pharmacokinetics.
Keywords: finasteride; food administration; pharmacogenetics; tadalafil.
Conflict of interest statement
Francisco Abad-Santos and Dolores Ochoa have been consultants or investigators in clinical trials sponsored by the following pharmaceutical companies: Abbott, Alter, Chemo, Cinfa, FAES, Farmalider, Ferrer, GlaxoSmithKline, Galenicum, Gilead, Italfarmaco, Janssen-Cilag, Kern, Moderna, MSD, Normon, Novartis, Servier, Silverpharma, Teva, and Zambon. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Bioequivalence and food effect of a fixed-dose combination of macitentan and tadalafil: Adaptive design in the COVID-19 pandemic.Pharmacol Res Perspect. 2021 Oct;9(5):e00846. doi: 10.1002/prp2.846. Pharmacol Res Perspect. 2021. PMID: 34624174 Free PMC article. Clinical Trial.
-
Bioequivalence of a single 10-mg dose of finasteride 5-mg oral disintegrating tablets and standard tablets in healthy adult male Han Chinese volunteers: a randomized sequence, open-label, two-way crossover study.Clin Ther. 2009 Oct;31(10):2242-8. doi: 10.1016/j.clinthera.2009.09.015. Clin Ther. 2009. PMID: 19922895 Clinical Trial.
-
Results From Phase I Studies Investigating the Dose Linearity of Finerenone Tablets and the Influence of Food or pH-Modifying Comedications on its Pharmacokinetics in Healthy Male Volunteers.Eur J Drug Metab Pharmacokinet. 2022 Jul;47(4):549-559. doi: 10.1007/s13318-022-00770-z. Epub 2022 May 25. Eur J Drug Metab Pharmacokinet. 2022. PMID: 35612708
-
Combination of tadalafil and finasteride for improving the symptoms of benign prostatic hyperplasia: critical appraisal and patient focus.Ther Clin Risk Manag. 2015 Mar 30;11:507-13. doi: 10.2147/TCRM.S80353. eCollection 2015. Ther Clin Risk Manag. 2015. PMID: 25848297 Free PMC article. Review.
-
Dihydrotestosterone and the concept of 5alpha-reductase inhibition in human benign prostatic hyperplasia.World J Urol. 2002 Apr;19(6):413-25. doi: 10.1007/s00345-002-0248-5. World J Urol. 2002. PMID: 12022710 Review.
Cited by
-
What do we really know about the external urethral sphincter?Continence (Amst). 2024 Jun;10:None. doi: 10.1016/j.cont.2024.101223. Continence (Amst). 2024. PMID: 39669761 Free PMC article. Review.
References
-
- Ng M., Baradhi K.M. StatPearls. StatPearls Publishing; Treasure Island, FL, USA: 2022. Benign Prostatic Hyperplasia. - PubMed
Grants and funding
- FI20/00090/Instituto de Salud Carlos III and European Social Fund
- ICI20/00131/Instituto de Salud Carlos III
- Margarita Salas contract/Autonomous University of Madrid
- FPI-UAM 2021/Autonomous University of Madrid
- 2022-C23.I01.P03.S0020-0000031 and 09-PIN1-00015.6/2022./Programa Investigo, European Union, NextGenerationEU funds of the Recovery and Resilience Facility
LinkOut - more resources
Full Text Sources
Miscellaneous