Vitamin D3 Exerts Beneficial Effects on C2C12 Myotubes through Activation of the Vitamin D Receptor (VDR)/Sirtuins (SIRT)1/3 Axis
- PMID: 38004107
- PMCID: PMC10674540
- DOI: 10.3390/nu15224714
Vitamin D3 Exerts Beneficial Effects on C2C12 Myotubes through Activation of the Vitamin D Receptor (VDR)/Sirtuins (SIRT)1/3 Axis
Abstract
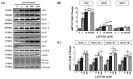
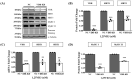
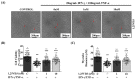
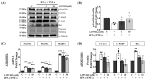
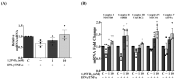
The onset of sarcopenia is associated with a decline in vitamin D receptor (VDR) expression, wherein reduced VDR levels contribute to muscle atrophy, while heightened expression promotes muscle hypertrophy. Like VDR, the age-related decline in protein deacetylase sirtuin (SIRT) expression is linked to the development of sarcopenia and age-related muscle dysfunction. This study aimed to investigate whether the VDR agonist 1,25-dihydroxyvitamin D3 (1,25VD3) exerts beneficial effects on muscles through interactions with sirtuins and, if so, the underlying molecular mechanisms. Treatment of 1,25VD3 in differentiating C2C12 myotubes substantially elevated VDR, SIRT1, and SIRT3 expression, enhancing their differentiation. Furthermore, 1,25VD3 significantly enhanced the expression of key myogenic markers, including myosin heavy chain (MyHC) proteins, MyoD, and MyoG, and increased the phosphorylation of AMPK and AKT. Conversely, VDR knockdown resulted in myotube atrophy and reduced SIRT1 and SIRT3 levels. In a muscle-wasting model triggered by IFN-γ/TNF-α in C2C12 myotubes, diminished VDR, SIRT1, and SIRT3 levels led to skeletal muscle atrophy and apoptosis. 1,25VD3 downregulated the increased expression of muscle atrophy-associated proteins, including FoxO3a, MAFbx, and MuRF1 in an IFN-γ/TNF-α induced atrophy model. Importantly, IFN-γ/TNF-α significantly reduced the mtDNA copy number in the C2C12 myotube, whereas the presence of 1,25VD3 effectively prevented this decrease. These results support that 1,25VD3 could serve as a potential preventive or therapeutic agent against age-related muscle atrophy by enhancing the VDR/SIRT1/SIRT3 axis.
Keywords: mitochondrial biogenesis; muscle atrophy; oxidative phosphorylation (OXPHOS); sarcopenia; sirtuins; vitamin D; vitamin D receptor (VDR).
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Differential effects of cholecalciferol and calcitriol on muscle proteolysis and oxidative stress in angiotensin II-induced C2C12 myotube atrophy.Physiol Rep. 2024 Apr;12(8):e16011. doi: 10.14814/phy2.16011. Physiol Rep. 2024. PMID: 38627219 Free PMC article.
-
Isoflavones derived from soy beans prevent MuRF1-mediated muscle atrophy in C2C12 myotubes through SIRT1 activation.J Nutr Sci Vitaminol (Tokyo). 2013;59(4):317-24. doi: 10.3177/jnsv.59.317. J Nutr Sci Vitaminol (Tokyo). 2013. PMID: 24064732
-
Vitamin D/Vitamin D Receptor Signaling Attenuates Skeletal Muscle Atrophy by Suppressing Renin-Angiotensin System.J Bone Miner Res. 2022 Jan;37(1):121-136. doi: 10.1002/jbmr.4441. Epub 2021 Sep 29. J Bone Miner Res. 2022. PMID: 34490953
-
The Vitamin D/Vitamin D receptor (VDR) axis in muscle atrophy and sarcopenia.Cell Signal. 2022 Aug;96:110355. doi: 10.1016/j.cellsig.2022.110355. Epub 2022 May 17. Cell Signal. 2022. PMID: 35595176 Review.
-
Vitamin D, muscle recovery, sarcopenia, cachexia, and muscle atrophy.Nutrition. 2019 Apr;60:66-69. doi: 10.1016/j.nut.2018.09.031. Epub 2018 Oct 7. Nutrition. 2019. PMID: 30529188 Review.
Cited by
-
The effect of Miya on skeletal muscle changes by regulating gut microbiota in rats with osteoarthritis through AMPK pathway.BMC Musculoskelet Disord. 2024 Dec 30;25(1):1081. doi: 10.1186/s12891-024-08203-5. BMC Musculoskelet Disord. 2024. PMID: 39736635 Free PMC article.
-
Effect of vitamin D supplementation on motor symptoms in Parkinson's disease: a meta-analysis of randomized controlled trials.Front Nutr. 2025 Jun 9;12:1500875. doi: 10.3389/fnut.2025.1500875. eCollection 2025. Front Nutr. 2025. PMID: 40551732 Free PMC article.
-
Danshensu sodium salt alleviates muscle atrophy via CaMKII-PGC1α-FoxO3a signaling pathway in D-galactose-induced models.FASEB J. 2025 Jan 31;39(2):e70280. doi: 10.1096/fj.202402158R. FASEB J. 2025. PMID: 39835720 Free PMC article.
-
Vitamin D and Sarcopenia in the Senior People: A Review of Mechanisms and Comprehensive Prevention and Treatment Strategies.Ther Clin Risk Manag. 2024 Sep 5;20:577-595. doi: 10.2147/TCRM.S471191. eCollection 2024. Ther Clin Risk Manag. 2024. PMID: 39253031 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

