Co-Delivery of a High Dose of Amphotericin B and Itraconazole by Means of a Dry Powder Inhaler Formulation for the Treatment of Severe Fungal Pulmonary Infections
- PMID: 38004579
- PMCID: PMC10675812
- DOI: 10.3390/pharmaceutics15112601
Co-Delivery of a High Dose of Amphotericin B and Itraconazole by Means of a Dry Powder Inhaler Formulation for the Treatment of Severe Fungal Pulmonary Infections
Abstract
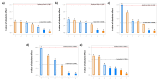
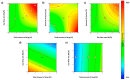
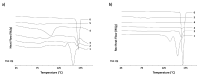
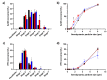
Over the past few decades, there has been a considerable rise in the incidence and prevalence of pulmonary fungal infections, creating a global health problem due to a lack of antifungal therapies specifically designed for pulmonary administration. Amphotericin B (AmB) and itraconazole (ITR) are two antifungal drugs with different mechanisms of action that have been widely employed in antimycotic therapy. In this work, microparticles containing a high dose of AmB and ITR (20, 30, and 40% total antifungal drug loading) were engineered for use in dry powder inhalers (DPIs) with an aim to improve the pharmacological effect, thereby enhancing the existing off-label choices for pulmonary administration. A Design of Experiment (DoE) approach was employed to prepare DPI formulations consisting of AmB-ITR encapsulated within γ-cyclodextrin (γ-CD) alongside functional excipients, such as mannitol and leucine. In vitro deposition indicated a favourable lung deposition pattern characterised by an upper ITR distribution (mass median aerodynamic diameter (MMAD) ~ 6 µm) along with a lower AmB deposition (MMAD ~ 3 µm). This offers significant advantages for treating fungal infections, not only in the lung parenchyma but also in the upper respiratory tract, considering that Aspergillus spp. can cause upper and lower airway disorders. The in vitro deposition profile of ITR and larger MMAD was related to the higher unencapsulated crystalline fraction of the drug, which may be altered using a higher concentration of γ-CD.
Keywords: amphotericin B; aspergillosis; dry powder inhaler; infection; itraconazole; lung deposition.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Targeting lung macrophages for fungal and parasitic pulmonary infections with innovative amphotericin B dry powder inhalers.Int J Pharm. 2023 Mar 25;635:122788. doi: 10.1016/j.ijpharm.2023.122788. Epub 2023 Mar 1. Int J Pharm. 2023. PMID: 36863544
-
Can amphotericin B and itraconazole be co-delivered orally? Tailoring oral fixed-dose combination coated granules for systemic mycoses.Eur J Pharm Biopharm. 2023 Feb;183:74-91. doi: 10.1016/j.ejpb.2023.01.003. Epub 2023 Jan 7. Eur J Pharm Biopharm. 2023. PMID: 36623752
-
Development of drug alone and carrier-based GLP-1 dry powder inhaler formulations.Int J Pharm. 2022 Apr 5;617:121601. doi: 10.1016/j.ijpharm.2022.121601. Epub 2022 Feb 16. Int J Pharm. 2022. PMID: 35181460
-
Liposomal amphotericin B: a review of its use as empirical therapy in febrile neutropenia and in the treatment of invasive fungal infections.Drugs. 2009;69(3):361-92. doi: 10.2165/00003495-200969030-00010. Drugs. 2009. PMID: 19275278 Review.
-
Inhaled Antifungal Agents for the Treatment and Prophylaxis of Pulmonary Mycoses.Curr Pharm Des. 2021;27(12):1453-1468. doi: 10.2174/1381612826666210101153547. Curr Pharm Des. 2021. PMID: 33388013 Review.
Cited by
-
A Novel Combined Dry Powder Inhaler Comprising Nanosized Ketoprofen-Embedded Mannitol-Coated Microparticles for Pulmonary Inflammations: Development, In Vitro-In Silico Characterization, and Cell Line Evaluation.Pharmaceuticals (Basel). 2024 Jan 7;17(1):75. doi: 10.3390/ph17010075. Pharmaceuticals (Basel). 2024. PMID: 38256908 Free PMC article.
-
Lipid-Based Nanotechnology: Liposome.Pharmaceutics. 2023 Dec 26;16(1):34. doi: 10.3390/pharmaceutics16010034. Pharmaceutics. 2023. PMID: 38258045 Free PMC article. Review.
-
Lung Delivery of Lactose-Free Microparticles Loaded with Azithromycin for the Treatment of Bacterial Infections.Pharmaceutics. 2025 Jun 11;17(6):770. doi: 10.3390/pharmaceutics17060770. Pharmaceutics. 2025. PMID: 40574082 Free PMC article.
References
-
- de Pablo E., O’Connell P., Fernández-García R., Marchand S., Chauzy A., Tewes F., Dea-Ayuela M.A., Kumar D., Bolás F., Ballesteros M.P., et al. Targeting lung macrophages for fungal and parasitic pulmonary infections with innovative amphotericin B dry powder inhalers. Int. J. Pharm. 2023;635:122788. doi: 10.1016/j.ijpharm.2023.122788. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

