Field-Induced Agglomerations of Polyethylene-Glycol-Functionalized Nanoclusters: Rheological Behaviour and Optical Microscopy
- PMID: 38004590
- PMCID: PMC10675764
- DOI: 10.3390/pharmaceutics15112612
Field-Induced Agglomerations of Polyethylene-Glycol-Functionalized Nanoclusters: Rheological Behaviour and Optical Microscopy
Abstract
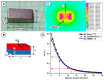
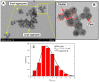
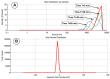
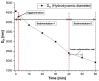
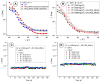
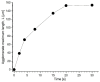
This research aims to investigate the agglomeration processes of magnetoresponsive functionalized nanocluster suspensions in a magnetic field, as well as how these structures impact the behaviour of these suspensions in biomedical applications. The synthesis, shape, colloidal stability, and magnetic characteristics of PEG-functionalized nanoclusters are described in this paper. Experiments using TEM, XPS, dynamic light scattering (DLS), VSM, and optical microscopy were performed to study chain-like agglomeration production and its influence on colloidal behaviour in physiologically relevant suspensions. The applied magnetic field aligns the magnetic moments of the nanoclusters. It provides an attraction between neighbouring particles, resulting in the formation of chains, linear aggregates, or agglomerates of clusters aligned along the applied field direction. Optical microscopy has been used to observe the creation of these aligned linear formations. The design of chain-like structures can cause considerable changes in the characteristics of ferrofluids, ranging from rheological differences to colloidal stability changes.
Keywords: chain formation; magnetic particle targeting; magnetoresponsive nanocomposite; magnetorheological properties; optical microscopy; particle aggregation/agglomeration.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






Similar articles
-
Magnetoresponsive Functionalized Nanocomposite Aggregation Kinetics and Chain Formation at the Targeted Site during Magnetic Targeting.Pharmaceutics. 2022 Sep 12;14(9):1923. doi: 10.3390/pharmaceutics14091923. Pharmaceutics. 2022. PMID: 36145671 Free PMC article.
-
The effect of magnetically induced linear aggregates on proton transverse relaxation rates of aqueous suspensions of polymer coated magnetic nanoparticles.Nanoscale. 2013 Mar 7;5(5):2152-63. doi: 10.1039/c3nr32979h. Epub 2013 Feb 6. Nanoscale. 2013. PMID: 23389324
-
Erratum: Preparation of Poly(pentafluorophenyl acrylate) Functionalized SiO2 Beads for Protein Purification.J Vis Exp. 2019 Apr 30;(146). doi: 10.3791/6328. J Vis Exp. 2019. PMID: 31038480
-
Chain Formation and Phase Separation in Ferrofluids: The Influence on Viscous Properties.Materials (Basel). 2020 Sep 7;13(18):3956. doi: 10.3390/ma13183956. Materials (Basel). 2020. PMID: 32906703 Free PMC article. Review.
-
Magnetic nanofluids (Ferrofluids): Recent advances, applications, challenges, and future directions.Adv Colloid Interface Sci. 2023 Jan;311:102810. doi: 10.1016/j.cis.2022.102810. Epub 2022 Nov 14. Adv Colloid Interface Sci. 2023. PMID: 36417827 Review.
References
-
- Douglas G., Van Kampen E., Hale A.B., McNeill E., Patel J., Crabtree M.J., Ali Z., Hoerr R.A., Alp N.J., Channon K.M. Endothelial cell repopulation after stenting determines in-stent neointima formation: Effects of bare-metal vs. drug-eluting stents and genetic endothelial cell modification. Eur. Heart J. 2012;34:3378–3388. doi: 10.1093/eurheartj/ehs240. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

