Photodynamic Therapy-Induced Anti-Tumor Immunity: Influence Factors and Synergistic Enhancement Strategies
- PMID: 38004595
- PMCID: PMC10675361
- DOI: 10.3390/pharmaceutics15112617
Photodynamic Therapy-Induced Anti-Tumor Immunity: Influence Factors and Synergistic Enhancement Strategies
Abstract
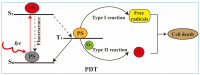
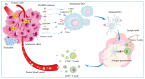
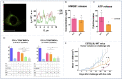
Photodynamic therapy (PDT) is an approved therapeutic procedure that exerts cytotoxic activity towards tumor cells by activating photosensitizers (PSs) with light exposure to produce reactive oxygen species (ROS). Compared to traditional treatment strategies such as surgery, chemotherapy, and radiation therapy, PDT not only kills the primary tumors, but also effectively suppresses metastatic tumors by activating the immune response. However, the anti-tumor immune effects induced by PDT are influenced by several factors, including the localization of PSs in cells, PSs concentration, fluence rate of light, oxygen concentration, and the integrity of immune function. In this review, we systematically summarize the influence factors of anti-tumor immune effects mediated by PDT. Furthermore, an update on the combination of PDT and other immunotherapy strategies are provided. Finally, the future directions and challenges of anti-tumor immunity induced by PDT are discussed.
Keywords: combination therapy; influence factors; innate immunity; photodynamic therapy; specific immunity.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Insight into the Prospects for Tumor Therapy Based on Photodynamic Immunotherapy.Pharmaceuticals (Basel). 2022 Nov 4;15(11):1359. doi: 10.3390/ph15111359. Pharmaceuticals (Basel). 2022. PMID: 36355531 Free PMC article. Review.
-
Combination of photodynamic therapy (PDT) and anti-tumor immunity in cancer therapy.J Pharm Investig. 2018;48(2):143-151. doi: 10.1007/s40005-017-0377-x. Epub 2018 Nov 1. J Pharm Investig. 2018. PMID: 30680248 Free PMC article. Review.
-
Aggregation-induced emission photosensitizer/bacteria biohybrids enhance Cerenkov radiation-induced photodynamic therapy by activating anti-tumor immunity for synergistic tumor treatment.Acta Biomater. 2023 Sep 1;167:519-533. doi: 10.1016/j.actbio.2023.06.009. Epub 2023 Jun 15. Acta Biomater. 2023. PMID: 37328041
-
Nano-photosensitizers for enhanced photodynamic therapy.Photodiagnosis Photodyn Ther. 2021 Dec;36:102597. doi: 10.1016/j.pdpdt.2021.102597. Epub 2021 Oct 24. Photodiagnosis Photodyn Ther. 2021. PMID: 34699982 Review.
-
Photodynamic Therapy Combined with Ferroptosis Is a Synergistic Antitumor Therapy Strategy.Cancers (Basel). 2023 Oct 19;15(20):5043. doi: 10.3390/cancers15205043. Cancers (Basel). 2023. PMID: 37894410 Free PMC article. Review.
Cited by
-
The Latest Look at PDT and Immune Checkpoints.Curr Issues Mol Biol. 2024 Jul 8;46(7):7239-7257. doi: 10.3390/cimb46070430. Curr Issues Mol Biol. 2024. PMID: 39057071 Free PMC article. Review.
-
Photodynamic Therapy Using IR-783 Liposomes for Advanced Tongue and Breast Cancers in Humans.J Funct Biomater. 2024 Dec 2;15(12):363. doi: 10.3390/jfb15120363. J Funct Biomater. 2024. PMID: 39728163 Free PMC article.
-
Photodynamic Therapy in Cancer: Insights into Cellular and Molecular Pathways.Curr Issues Mol Biol. 2025 Jan 21;47(2):69. doi: 10.3390/cimb47020069. Curr Issues Mol Biol. 2025. PMID: 39996790 Free PMC article. Review.
-
Advances in Nanodynamic Therapy for Cancer Treatment.Nanomaterials (Basel). 2024 Apr 8;14(7):648. doi: 10.3390/nano14070648. Nanomaterials (Basel). 2024. PMID: 38607182 Free PMC article. Review.
-
Functionalized and Theranostic Lipidic and Tocosomal Drug Delivery Systems: Potentials and Limitations in Cancer Photodynamic Therapy.Adv Pharm Bull. 2024 Oct;14(3):524-536. doi: 10.34172/apb.2024.038. Epub 2024 Mar 11. Adv Pharm Bull. 2024. PMID: 39494248 Free PMC article. Review.
References
-
- Togsverd-Bo K., Sandberg C., Helsing P., Mørk G., Wennberg A.M., Wulf H.C., Haedersdal M. Cyclic photodynamic therapy delays first onset of actinic keratoses in renal transplant recipients: A 5-year randomized controlled trial with 12-month follow-up. J. Eur. Acad. Dermatol. Venereol. 2022;36:E946–E948. doi: 10.1111/jdv.18374. - DOI - PubMed
-
- Barreca M., Ingarra A.M., Raimondi M.V., Spanò V., Piccionello A.P., De Franco M., Menilli L., Gandin V., Miolo G., Barraja P., et al. New tricyclic systems as photosensitizers towards triple negative breast cancer cells. Arch. Pharmacal Res. 2022;45:806–821. doi: 10.1007/s12272-022-01414-1. - DOI - PMC - PubMed
Publication types
Grants and funding
- 2022YFA1207600/the National Key R&D Program of China
- T2293750/the Major Program of the National Natural Science Foundation of China
- T2293753/the Major Program of the National Natural Science Foundation of China
- XSQD-202023003/the Beijing Institute of Technology Research Fund Program for Young Scholars
- 2019-I2M-5-061/CAMS Innovation Fund for Medical Sciences (CIFMS)
LinkOut - more resources
Full Text Sources

