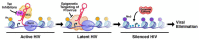HIV-1 Remission: Accelerating the Path to Permanent HIV-1 Silencing
- PMID: 38005849
- PMCID: PMC10674359
- DOI: 10.3390/v15112171
HIV-1 Remission: Accelerating the Path to Permanent HIV-1 Silencing
Abstract
Despite remarkable progress, a cure for HIV-1 infection remains elusive. Rebound competent latent and transcriptionally active reservoir cells persevere despite antiretroviral therapy and rekindle infection due to inefficient proviral silencing. We propose a novel "block-lock-stop" approach, entailing long term durable silencing of viral expression towards an irreversible transcriptionally inactive latent provirus to achieve long term antiretroviral free control of the virus. A graded transformation of remnant HIV-1 in PLWH from persistent into silent to permanently defective proviruses is proposed, emulating and accelerating the natural path that human endogenous retroviruses (HERVs) take over millions of years. This hypothesis was based on research into delineating the mechanisms of HIV-1 latency, lessons from latency reversing agents and advances of Tat inhibitors, as well as expertise in the biology of HERVs. Insights from elite controllers and the availability of advanced genome engineering technologies for the direct excision of remnant virus set the stage for a rapid path to an HIV-1 cure.
Keywords: HERV; HIV-1 cure; latency; transcriptional silencing.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Oomen P.G.A., Dijkstra S., Hofstra L.M., Nijhuis M.M., Verbon A., Mudrikova T., Wensing A.M.J., Hoepelman A.I.M., Van Welzen B.J. Integrated analysis of viral blips, residual viremia, and associated factors in people with HIV: Results from a retrospective cohort study. J. Med. Virol. 2023;95:e29178. doi: 10.1002/jmv.29178. - DOI - PubMed
-
- Singh K., Natarajan V., Dewar R., Rupert A., Badralmaa Y., Zhai T., Winchester N., Scrimieri F., Smith M., Davis I., et al. Long-term persistence of transcriptionally active ‘defective’ HIV-1 proviruses: Implications for persistent immune activation during antiretroviral therapy. AIDS. 2023;37:2119–2130. doi: 10.1097/QAD.0000000000003667. - DOI - PMC - PubMed
-
- Mavigner M., Delobel P., Cazabat M., Dubois M., L’Faqihi-Olive F.E., Raymond S., Pasquier C., Marchou B., Massip P., Izopet J. HIV-1 residual viremia correlates with persistent T-cell activation in poor immunological responders to combination antiretroviral therapy. PLoS ONE. 2009;4:e7658. doi: 10.1371/journal.pone.0007658. - DOI - PMC - PubMed
MeSH terms
Grants and funding
- P01 AI169606/AI/NIAID NIH HHS/United States
- UM1 AI164559/AI/NIAID NIH HHS/United States
- R01 DA052027/DA/NIDA NIH HHS/United States
- R21 AI172060/AI/NIAID NIH HHS/United States
- R01 AI167732/AI/NIAID NIH HHS/United States
- R01 CA260691/CA/NCI NIH HHS/United States
- R37 AI083139/AI/NIAID NIH HHS/United States
- K01 OD031900/OD/NIH HHS/United States
- R37 AI165137/AI/NIAID NIH HHS/United States
- R21 AI170166/AI/NIAID NIH HHS/United States
- P30 AI036214/AI/NIAID NIH HHS/United States
- R01 DK131526/DK/NIDDK NIH HHS/United States
- R01 AI147777/AI/NIAID NIH HHS/United States