Comparative Immune Response after Vaccination with SOBERANA® 02 and SOBERANA® plus Heterologous Scheme and Natural Infection in Young Children
- PMID: 38005968
- PMCID: PMC10675375
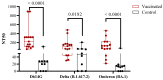
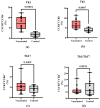
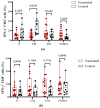
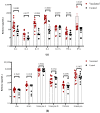
- DOI: 10.3390/vaccines11111636
Comparative Immune Response after Vaccination with SOBERANA® 02 and SOBERANA® plus Heterologous Scheme and Natural Infection in Young Children
Abstract
(1) Background: In children, SARS-CoV-2 infection is mostly accompanied by mild COVID-19 symptoms. However, multisystem inflammatory syndrome (MIS-C) and long-term sequelae are often severe complications. Therefore, the protection of the pediatric population against SARS-CoV-2 with effective vaccines is particularly important. Here, we compare the humoral and cellular immune responses elicited in children (n = 15, aged 5-11 years) vaccinated with the RBD-based vaccines SOBERANA® 02 and SOBERANA® Plus combined in a heterologous scheme with those from children (n = 10, aged 4-11 years) who recovered from mild symptomatic COVID-19. (2) Methods: Blood samples were taken 14 days after the last dose for vaccinated children and 45-60 days after the infection diagnosis for COVID-19 recovered children. Anti-RBD IgG and ACE2-RBD inhibition were assessed by ELISA; IgA, cytokines, and cytotoxic-related proteins were determined by multiplex assays. Total B and T cell subpopulations and IFN-γ release were measured by multiparametric flow cytometry using a large panel of antibodies after in vitro stimulation with S1 peptides. (3) Results: Significant higher levels of specific anti-RBD IgG and IgA and ACE2-RBD inhibition capacity were found in vaccinated children in comparison to COVID-19 recovered children. Th1-like and Th2-like CD4+ T cells were also significantly higher in vaccinated subjects. IFN-γ secretion was higher in central memory CD4+ T cells of COVID-19 recovered children, but no differences between both groups were found in the CD4+ and CD8+ T cell effector, terminal effector, and naïve T cell subpopulations. In contrast to low levels of IL-4, high levels of IL-2, IL-6, IFN-γ, and IL-10 suggest a predominant Th1 cell polarization. Cytotoxic-related proteins granzyme A and B, perforin, and granulin were also found in the supernatant after S1 stimulation in both vaccinated and recovered children. (4) Conclusions: Vaccination with the heterologous scheme of SOBERANA® 02/SOBERANA® Plus induces a stronger antibody and cellular immune response compared to natural infections in young children.
Keywords: COVID-19; SARS-CoV-2; SARS-CoV-2 RBD IgG; children; memory T cell; vaccine.
Conflict of interest statement
The authors R.P.-N., L.M.R.-N., B.P.-M., M.R.-G., M.G.-F., I.P.-Á., A.G.-H., Y.V.-B., D.G.-R. and V.V.-B. declare to be employees at Finlay Vaccine Institute.
Figures


Similar articles
-
Safety and durability of the immune response after vaccination with the heterologous schedule of anti-COVID-19 vaccines SOBERANA®02 and SOBERANA® Plus in children 3-18 years old.Vaccine X. 2024 Dec 6;22:100595. doi: 10.1016/j.jvacx.2024.100595. eCollection 2025 Jan. Vaccine X. 2024. PMID: 39737224 Free PMC article.
-
Real-world effectiveness of the heterologous SOBERANA-02 and SOBERANA-Plus vaccine scheme in 2-11 years-old children during the SARS-CoV-2 Omicron wave in Cuba: a longitudinal case-population study.Lancet Reg Health Am. 2024 Apr 23;34:100750. doi: 10.1016/j.lana.2024.100750. eCollection 2024 Jun. Lancet Reg Health Am. 2024. PMID: 38699214 Free PMC article.
-
Safety and immunogenicity of anti-SARS CoV-2 vaccine SOBERANA 02 in homologous or heterologous scheme: Open label phase I and phase IIa clinical trials.Vaccine. 2022 Jul 29;40(31):4220-4230. doi: 10.1016/j.vaccine.2022.05.082. Epub 2022 Jun 6. Vaccine. 2022. PMID: 35691871 Free PMC article. Clinical Trial.
-
Safety and efficacy of the two doses conjugated protein-based SOBERANA-02 COVID-19 vaccine and of a heterologous three-dose combination with SOBERANA-Plus: a double-blind, randomised, placebo-controlled phase 3 clinical trial.Lancet Reg Health Am. 2023 Feb;18:100423. doi: 10.1016/j.lana.2022.100423. Epub 2022 Dec 31. Lancet Reg Health Am. 2023. PMID: 36618081 Free PMC article.
-
Safety and immunogenicity of anti-SARS-CoV-2 heterologous scheme with SOBERANA 02 and SOBERANA Plus vaccines: Phase IIb clinical trial in adults.Med. 2022 Nov 11;3(11):760-773.e5. doi: 10.1016/j.medj.2022.08.001. Epub 2022 Aug 8. Med. 2022. PMID: 35998623 Free PMC article. Clinical Trial.
Cited by
-
Safety and durability of the immune response after vaccination with the heterologous schedule of anti-COVID-19 vaccines SOBERANA®02 and SOBERANA® Plus in children 3-18 years old.Vaccine X. 2024 Dec 6;22:100595. doi: 10.1016/j.jvacx.2024.100595. eCollection 2025 Jan. Vaccine X. 2024. PMID: 39737224 Free PMC article.
-
Functional T cell response to COVID-19 vaccination with or without natural infection with SARS-CoV-2 in adults and children.Sci Rep. 2025 Apr 17;15(1):13341. doi: 10.1038/s41598-025-95870-6. Sci Rep. 2025. PMID: 40247005 Free PMC article.
-
Real-world effectiveness of the heterologous SOBERANA-02 and SOBERANA-Plus vaccine scheme in 2-11 years-old children during the SARS-CoV-2 Omicron wave in Cuba: a longitudinal case-population study.Lancet Reg Health Am. 2024 Apr 23;34:100750. doi: 10.1016/j.lana.2024.100750. eCollection 2024 Jun. Lancet Reg Health Am. 2024. PMID: 38699214 Free PMC article.
-
Beyond interferon gamma - decreased cellular response to COVID-19 vaccination booster in patients with autoimmune inflammatory rheumatic diseases.Front Immunol. 2025 Mar 28;16:1568439. doi: 10.3389/fimmu.2025.1568439. eCollection 2025. Front Immunol. 2025. PMID: 40226625 Free PMC article.
References
-
- Martin B., DeWitt P.E., Russell S., Anand A., Bradwell K.R., Bremer C., Gabriel D., Girvin A.T., Hajagos J.G., McMurry J.A., et al. Characteristics, Outcomes, and Severity Risk Factors Associated with SARS-CoV-2 Infection among Children in the US National COVID Cohort Collaborative. JAMA Netw. Open. 2022;5:e2143151. doi: 10.1001/jamanetworkopen.2021.43151. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

