An Evaluation of Urease A Subunit Nanocapsules as a Vaccine in a Mouse Model of Helicobacter pylori Infection
- PMID: 38005984
- PMCID: PMC10674275
- DOI: 10.3390/vaccines11111652
An Evaluation of Urease A Subunit Nanocapsules as a Vaccine in a Mouse Model of Helicobacter pylori Infection
Abstract
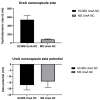
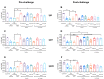
Using removable silica templates, protein nanocapsules comprising the A subunit of Helicobacter pylori urease (UreA) were synthesised. The templates were of two sizes, with solid core mesoporous shell (SC/MS) silica templates giving rise to nanocapsules of average diameter 510 nm and mesoporous (MS) silica templates giving rise to nanocapsules of average diameter 47 nm. Both were shown to be highly monodispersed and relatively homogenous in structure. Various combinations of the nanocapsules in formulation were assessed as vaccines in a mouse model of H. pylori infection. Immune responses were evaluated and protective efficacy assessed. It was demonstrated that vaccination of mice with the larger nanocapsules combined with an adjuvant was able to significantly reduce colonisation.
Keywords: Helicobacter pylori; nanocapsule; silica nanoparticle template; urease alpha subunit; vaccination.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Solid Lipid Nanoparticles Delivering a DNA Vaccine Encoding Helicobacter pylori Urease A Subunit: Immune Analyses before and after a Mouse Model of Infection.Int J Mol Sci. 2024 Jan 16;25(2):1076. doi: 10.3390/ijms25021076. Int J Mol Sci. 2024. PMID: 38256149 Free PMC article.
-
Protection Against Helicobacter pylori Infection in BALB/c Mouse Model by Oral Administration of Multivalent Epitope-Based Vaccine of Cholera Toxin B Subunit-HUUC.Front Immunol. 2018 May 8;9:1003. doi: 10.3389/fimmu.2018.01003. eCollection 2018. Front Immunol. 2018. PMID: 29867978 Free PMC article.
-
Protein-only nanocapsules induce cross-presentation in dendritic cells, demonstrating potential as an antigen delivery system.Nanomedicine. 2020 Aug;28:102234. doi: 10.1016/j.nano.2020.102234. Epub 2020 Jun 11. Nanomedicine. 2020. PMID: 32522709
-
Perspectives from recent advances of Helicobacter pylori vaccines research.Helicobacter. 2022 Dec;27(6):e12926. doi: 10.1111/hel.12926. Epub 2022 Sep 22. Helicobacter. 2022. PMID: 36134470 Review.
-
The current status of Helicobacter pylori vaccines: a review.Helicobacter. 2007 Apr;12(2):89-102. doi: 10.1111/j.1523-5378.2007.00478.x. Helicobacter. 2007. PMID: 17309745 Review.
Cited by
-
Nanocapsules Comprised of Purified Protein: Construction and Applications in Vaccine Research.Vaccines (Basel). 2024 Apr 12;12(4):410. doi: 10.3390/vaccines12040410. Vaccines (Basel). 2024. PMID: 38675791 Free PMC article.
-
Bibliometric analysis of Helicobacter pylori vaccine development from 1993 to 2023.Front Microbiol. 2025 Mar 17;16:1479195. doi: 10.3389/fmicb.2025.1479195. eCollection 2025. Front Microbiol. 2025. PMID: 40165784 Free PMC article.
-
Developing a potent vaccine against Helicobacter pylori: critical considerations and challenges.Expert Rev Mol Med. 2024 Nov 25;27:e12. doi: 10.1017/erm.2024.19. Expert Rev Mol Med. 2024. PMID: 39584502 Free PMC article. Review.
-
Special Issue "Recent Advances in Nanoparticles in Molecular Biology".Int J Mol Sci. 2025 Jun 30;26(13):6321. doi: 10.3390/ijms26136321. Int J Mol Sci. 2025. PMID: 40650098 Free PMC article.
-
Solid Lipid Nanoparticles Delivering a DNA Vaccine Encoding Helicobacter pylori Urease A Subunit: Immune Analyses before and after a Mouse Model of Infection.Int J Mol Sci. 2024 Jan 16;25(2):1076. doi: 10.3390/ijms25021076. Int J Mol Sci. 2024. PMID: 38256149 Free PMC article.
References
LinkOut - more resources
Full Text Sources

