Recombinant cellular model system for human muscle-type nicotinic acetylcholine receptor α12β1δε
- PMID: 38006565
- PMCID: PMC10746606
- DOI: 10.1007/s12192-023-01395-0
Recombinant cellular model system for human muscle-type nicotinic acetylcholine receptor α12β1δε
Abstract
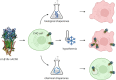
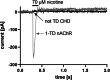
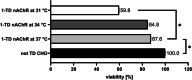
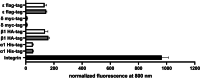
The human muscle-type nicotinic acetylcholine receptor α12β1δε (nAChR) is a complex transmembrane receptor needed for drug screening for disorders like congenital myasthenic syndromes and multiple pterygium syndrome. Until today, most models are still using the nAChR from Torpedo californica electric ray. A simple reproducible cellular system expressing functional human muscle-type nAChR is still missing. This study addressed this issue and further tested the hypothesis that different chaperones, both biological and chemical, and posttranslational modification supporting substances as well as hypothermic incubation are able to increase the nAChR yield. Therefore, Gibson cloning was used to generate transfer plasmids carrying the sequence of nAChR or chosen biological chaperones to support the nAChR folding in the cellular host. Viral transduction was used for stable integration of these transgenes in Chinese hamster ovary cells (CHO). Proteins were detected with Western blot, in-cell and on-cell Western, and the function of the receptor with voltage clamp analysis. We show that the internalization of nAChR into plasma membranes was sufficient for detection and function. Additional transgenic overexpression of biological chaperones did result in a reduced nAChR expression. Chemical chaperones, posttranslational modification supporting substances, and hypothermic conditions are well-suited supporting applications to increase the protein levels of different subunits. This study presents a stable and functional cell line that expresses human muscle-type nAChR and yields can be further increased using the chemical chaperone nicotine without affecting cell viability. The simplified access to this model system should enable numerous applications beyond drug development. Graphical abstract created with http://biorender.com.
Keywords: Molecular chaperones; Nicotine; Nicotinic acetylcholine receptor.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
N-Glycosylation Deficiency in Transgene α7 nAChR and RIC3 Expressing CHO Cells Without NACHO.J Membr Biol. 2024 Aug;257(3-4):245-256. doi: 10.1007/s00232-024-00317-0. Epub 2024 Jul 5. J Membr Biol. 2024. PMID: 38967800
-
Functional properties of alpha7 nicotinic acetylcholine receptors co-expressed with RIC-3 in a stable recombinant CHO-K1 cell line.Assay Drug Dev Technol. 2008 Apr;6(2):181-93. doi: 10.1089/adt.2007.120. Assay Drug Dev Technol. 2008. PMID: 18471073
-
Nicotine activates cell-signaling pathways through muscle-type and neuronal nicotinic acetylcholine receptors in non-small cell lung cancer cells.Pulm Pharmacol Ther. 2007;20(6):629-41. doi: 10.1016/j.pupt.2006.07.001. Epub 2006 Aug 18. Pulm Pharmacol Ther. 2007. PMID: 17015027
-
A Review on the Receptor-ligand Molecular Interactions in the Nicotinic Receptor Signaling Systems.Pak J Biol Sci. 2018;21(2):51-66. doi: 10.3923/pjbs.2018.51.66. Pak J Biol Sci. 2018. PMID: 30221881 Review.
-
Nicotinic Acetylcholine Receptor Ligands, Cognitive Function, and Preclinical Approaches to Drug Discovery.Nicotine Tob Res. 2019 Feb 18;21(3):383-394. doi: 10.1093/ntr/nty166. Nicotine Tob Res. 2019. PMID: 30137518 Free PMC article. Review.
Cited by
-
N-Glycosylation Deficiency in Transgene α7 nAChR and RIC3 Expressing CHO Cells Without NACHO.J Membr Biol. 2024 Aug;257(3-4):245-256. doi: 10.1007/s00232-024-00317-0. Epub 2024 Jul 5. J Membr Biol. 2024. PMID: 38967800
-
Cell-Sonar, a Novel Method for Intracellular Tracking of Secretory Pathways.Cells. 2024 Aug 29;13(17):1449. doi: 10.3390/cells13171449. Cells. 2024. PMID: 39273021 Free PMC article.
-
Protein networking: nicotinic acetylcholine receptors and their protein-protein-associations.Mol Cell Biochem. 2024 Jul;479(7):1627-1642. doi: 10.1007/s11010-024-05032-x. Epub 2024 May 21. Mol Cell Biochem. 2024. PMID: 38771378 Review.
-
Autoimmune mechanisms elucidated through muscle acetylcholine receptor structures.Cell. 2025 May 1;188(9):2390-2406.e20. doi: 10.1016/j.cell.2025.03.004. Epub 2025 Apr 8. Cell. 2025. PMID: 40203823
-
Cell-Sonar, an Easy and Low-cost Method to Track a Target Protein by Expression Changes of Specific Protein Markers.Bio Protoc. 2025 Feb 5;15(3):e5206. doi: 10.21769/BioProtoc.5206. eCollection 2025 Feb 5. Bio Protoc. 2025. PMID: 39959296 Free PMC article.
References
-
- Flores CM, Rogers SW, Pabreza LA, Wolfe BB, Kellar KJ. A subtype of nicotinic cholinergic receptor in rat brain is composed of alpha 4 and beta 2 subunits and is up-regulated by chronic nicotine treatment. Mol Pharmacol. 1992;41:31–37. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

