Exploring the oral-gut linkage: Interrelationship between oral and systemic diseases
- PMID: 38007003
- PMCID: PMC11222583
- DOI: 10.1016/j.mucimm.2023.11.006
Exploring the oral-gut linkage: Interrelationship between oral and systemic diseases
Abstract
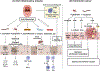
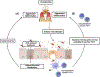
The oral cavity harbors a diverse microbiota that plays a significant role in maintaining homeostasis. Disruption of this balance can lead to various oral diseases, including periodontitis. Accumulating evidence suggests a connection between periodontitis and extra-oral diseases such as cardiovascular disease, rheumatoid arthritis, obesity, and diabetes. During periodontitis, oral bacteria enter the bloodstream directly, impacting extra-oral organs. Furthermore, recent studies have uncovered another pathway, the direct oral-gut axis, where oral bacteria translocate to the gut through an enteral route, influencing gut microbiota and metabolism. Oral pathobionts associated with exacerbation of periodontal disease are implicated in gut pathology, including inflammatory bowel disease and colorectal cancer through ectopic gut colonization. Furthermore, oral bacteria can provoke host immune responses, leading to colitis and other inflammatory diseases. Conversely, mechanisms by which extra-oral conditions exacerbate oral diseases, such as periodontitis, are also beginning to be elucidated. This review discusses the bidirectional interrelationship between oral and systemic diseases based on the oral-gut linkage.
Copyright © 2023 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Disclosures:
The authors declare no competing interests.
Figures
References
-
- Bahekar AA, Singh S, Saha S, Molnar J, Arora R The prevalence and incidence of coronary heart disease is significantly increased in periodontitis: a meta-analysis. American heart journal. 154, 830–837 (2007). - PubMed
-
- De Pablo P, Dietrich T, McAlindon TE Association of periodontal disease and tooth loss with rheumatoid arthritis in the US population. The Journal of rheumatology. 35, 70–76 (2008). - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

