Estradiol-mediated small GTP-binding protein GDP dissociation stimulator induction contributes to sex differences in resilience to ferroptosis in takotsubo syndrome
- PMID: 38007983
- PMCID: PMC10719533
- DOI: 10.1016/j.redox.2023.102961
Estradiol-mediated small GTP-binding protein GDP dissociation stimulator induction contributes to sex differences in resilience to ferroptosis in takotsubo syndrome
Abstract
Background: Declining beneficial cardiovascular actions of estradiol (E2) have been associated with disproportionate susceptibility to takotsubo syndrome (TTS) in postmenopausal women. However, the underlying mechanisms between E2 and this marked disproportion remain unclear. SmgGDS (small GTP-binding protein GDP dissociation stimulator), as a key modulator of cardiovascular disease, plays protective roles in reducing oxidative stress and exerts pleiotropic effects of statins. Whether SmgGDS levels are influenced by E2 status and the effect of SmgGDS on sex differences in TTS are poorly understood.
Methods: Clinical data were reviewed from TTS inpatients. Echocardiography, immunofluorescence, and immunohistochemistry were performed together with expression analysis to uncover phenotypic and mechanism changes in sex differences in TTS-like wild-type (WT) and SmgGDS± mice. HL-1 cardiomyocytes were used to further examine and validate molecular mechanisms.
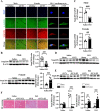
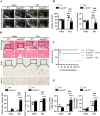
Results: In 14 TTS inpatients, TTS had a higher incidence in postmenopausal women as compared to premenopausal women and men. In murine TTS, female WT mice exhibited higher cardiac SmgGDS levels than male WT mice. Ovariectomy reduced SmgGDS expression in female WT mice similar to that observed in male mice, whereas E2 replacement in these ovariectomized (OVX) female mice reversed this effect. The physiological importance of this sex-specific E2-mediated SmgGDS response is underscored by the disparity in cardiac adaptation to isoproterenol (ISO) stimulation between both sexes of WT mice. E2-mediated SmgGDS induction conferred female protection against TTS-like acute cardiac injury involving ferritinophagy-mediated ferroptosis. No such cardioprotection was observed in male WT mice and OVX female. A causal role for SmgGDS in this sex-specific cardioprotective adaptation was indicated, inasmuch as SmgGDS deficiency abolished E2-modulated cardioprotection against ferritinophagy and aggravates TTS progression in both sexes. Consistently, knockdown of SmgGDS in HL-1 cardiomyocytes exacerbated ferroptosis in a ferritinophagy-dependent manner and abrogated the protective role of E2 against ferritinophagy. Mechanistically, our findings revealed that SmgGDS regulated E2-dependent cardioprotective effects via AMPK/mTOR signaling pathway. SmgGDS deficiency abolished E2-conferred protection against ferritinophagy through activating AMPK/mTOR pathway, while treatment with recombinant SmgGDS in HL-1 cells significantly mitigated this pathway-associated ferritinophagy activity.
Conclusions: These results demonstrate that SmgGDS is a central mediator of E2-conferred female cardioprotection against ferritinophagy-mediated ferroptosis in TTS.
Keywords: Ferritinophagy; Ferroptosis; Hormones; Sex; Takotsubo cardiomyopathy.
Copyright © 2023 The Authors. Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures






Similar articles
-
Statins exert the pleiotropic effects through small GTP-binding protein dissociation stimulator upregulation with a resultant Rac1 degradation.Arterioscler Thromb Vasc Biol. 2013 Jul;33(7):1591-600. doi: 10.1161/ATVBAHA.112.300922. Epub 2013 May 2. Arterioscler Thromb Vasc Biol. 2013. PMID: 23640485 Free PMC article. Clinical Trial.
-
SmgGDS as a Crucial Mediator of the Inhibitory Effects of Statins on Cardiac Hypertrophy and Fibrosis: Novel Mechanism of the Pleiotropic Effects of Statins.Hypertension. 2016 May;67(5):878-89. doi: 10.1161/HYPERTENSIONAHA.115.07089. Epub 2016 Mar 14. Hypertension. 2016. PMID: 26975711
-
Small GTP-Binding Protein GDP Dissociation Stimulator Prevents Thoracic Aortic Aneurysm Formation and Rupture by Phenotypic Preservation of Aortic Smooth Muscle Cells.Circulation. 2018 Nov 20;138(21):2413-2433. doi: 10.1161/CIRCULATIONAHA.118.035648. Circulation. 2018. PMID: 29921611
-
Sex Differences in Takotsubo Syndrome: A Narrative Review.J Womens Health (Larchmt). 2020 Aug;29(8):1122-1130. doi: 10.1089/jwh.2019.7741. Epub 2019 Sep 24. J Womens Health (Larchmt). 2020. PMID: 31549884 Review.
-
Takotsubo Syndrome and Oxidative Stress: Physiopathological Linkage and Future Perspectives.Antioxidants (Basel). 2025 Apr 27;14(5):522. doi: 10.3390/antiox14050522. Antioxidants (Basel). 2025. PMID: 40427405 Free PMC article. Review.
Cited by
-
SGLT2i continuously prevents cardiac hypertrophy by reducing ferroptosis via AMPK up-regulation.Mol Cell Biochem. 2025 Aug;480(8):4849-4864. doi: 10.1007/s11010-025-05294-z. Epub 2025 Apr 26. Mol Cell Biochem. 2025. PMID: 40285818
-
The Genetic Puzzle of the Stress-Induced Cardiomyopathy (Takotsubo Syndrome): State of Art and Future Perspectives.Biomolecules. 2025 Jun 24;15(7):926. doi: 10.3390/biom15070926. Biomolecules. 2025. PMID: 40723798 Free PMC article. Review.
-
Nrf2/UBE3B protects against acute lung injury by inhibiting ferritinophagy through the ubiquitination of NCOA4.Biol Direct. 2025 Jul 16;20(1):85. doi: 10.1186/s13062-025-00678-z. Biol Direct. 2025. PMID: 40671136 Free PMC article.
-
Nicotinamide Phosphoribosyltransferase Acetylation Mediating Muscle Dysfunction Contributes to Sleep Apnoea in Obesity.J Cachexia Sarcopenia Muscle. 2025 Feb;16(1):e13693. doi: 10.1002/jcsm.13693. J Cachexia Sarcopenia Muscle. 2025. PMID: 39901373 Free PMC article.
-
The Role of Ferroptosis in Women's Health and Diseases.MedComm (2020). 2025 Aug 15;6(8):e70296. doi: 10.1002/mco2.70296. eCollection 2025 Aug. MedComm (2020). 2025. PMID: 40821694 Free PMC article. Review.
References
-
- Ghadri J.R., Wittstein I.S., Prasad A., Sharkey S., Dote K., Akashi Y.J., Cammann V.L., Crea F., Galiuto L., Desmet W., et al. International expert consensus document on takotsubo syndrome (Part I): clinical characteristics, diagnostic criteria, and pathophysiology. Eur. Heart J. 2018;39(22):2032–2046. doi: 10.1093/eurheartj/ehy076. - DOI - PMC - PubMed
-
- Ghadri J.R., Wittstein I.S., Prasad A., Sharkey S., Dote K., Akashi Y.J., Cammann V.L., Crea F., Galiuto L., Desmet W., et al. International expert consensus document on takotsubo syndrome (Part II): diagnostic workup, outcome, and management. Eur. Heart J. 2018;39(22):2047–2062. doi: 10.1093/eurheartj/ehy077. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

