Listen to Your Gut: Key Concepts for Bioengineering Advanced Models of the Intestine
- PMID: 38009508
- PMCID: PMC10837392
- DOI: 10.1002/advs.202302165
Listen to Your Gut: Key Concepts for Bioengineering Advanced Models of the Intestine
Abstract
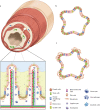
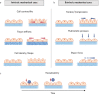
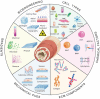
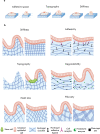
The intestine performs functions central to human health by breaking down food and absorbing nutrients while maintaining a selective barrier against the intestinal microbiome. Key to this barrier function are the combined efforts of lumen-lining specialized intestinal epithelial cells, and the supportive underlying immune cell-rich stromal tissue. The discovery that the intestinal epithelium can be reproduced in vitro as intestinal organoids introduced a new way to understand intestinal development, homeostasis, and disease. However, organoids reflect the intestinal epithelium in isolation whereas the underlying tissue also contains myriad cell types and impressive chemical and structural complexity. This review dissects the cellular and matrix components of the intestine and discusses strategies to replicate them in vitro using principles drawing from bottom-up biological self-organization and top-down bioengineering. It also covers the cellular, biochemical and biophysical features of the intestinal microenvironment and how these can be replicated in vitro by combining strategies from organoid biology with materials science. Particularly accessible chemistries that mimic the native extracellular matrix are discussed, and bioengineering approaches that aim to overcome limitations in modelling the intestine are critically evaluated. Finally, the review considers how further advances may extend the applications of intestinal models and their suitability for clinical therapies.
Keywords: biomaterials; disease modelling; intestine; organ-on-chip; organoids.
© 2023 The Authors. Advanced Science published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
A Novel Microphysiological Colon Platform to Decipher Mechanisms Driving Human Intestinal Permeability.Cell Mol Gastroenterol Hepatol. 2021;12(5):1719-1741. doi: 10.1016/j.jcmgh.2021.07.004. Epub 2021 Jul 17. Cell Mol Gastroenterol Hepatol. 2021. PMID: 34284165 Free PMC article.
-
Combining Human Organoids and Organ-on-a-Chip Technology to Model Intestinal Region-Specific Functionality.J Vis Exp. 2022 May 5;(183). doi: 10.3791/63724. J Vis Exp. 2022. PMID: 35604153
-
Biomaterials and bioengineering to guide tissue morphogenesis in epithelial organoids.Front Bioeng Biotechnol. 2022 Nov 17;10:1038277. doi: 10.3389/fbioe.2022.1038277. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 36466337 Free PMC article. Review.
-
Organoids as a tool to study homeostatic and pathological immune-epithelial interactions in the gut.Clin Exp Immunol. 2024 Sep 16;218(1):28-39. doi: 10.1093/cei/uxad118. Clin Exp Immunol. 2024. PMID: 38551817 Free PMC article. Review.
-
3D bioengineered tissue model of the large intestine to study inflammatory bowel disease.Biomaterials. 2019 Dec;225:119517. doi: 10.1016/j.biomaterials.2019.119517. Epub 2019 Sep 25. Biomaterials. 2019. PMID: 31580968 Free PMC article.
Cited by
-
Dynamics of Spatial Organization of Bacterial Communities in a Tunable Flow Gut Microbiome-on-a-Chip.Small. 2025 May;21(20):e2410258. doi: 10.1002/smll.202410258. Epub 2025 Apr 9. Small. 2025. PMID: 40201941 Free PMC article.
-
The Role of Gut Microbiome in Irritable Bowel Syndrome: Implications for Clinical Therapeutics.Biomolecules. 2024 Dec 21;14(12):1643. doi: 10.3390/biom14121643. Biomolecules. 2024. PMID: 39766350 Free PMC article. Review.
-
Long-range organization of intestinal 2D-crypts using exogenous Wnt3a micropatterning.Nat Commun. 2025 Jan 3;16(1):382. doi: 10.1038/s41467-024-55651-7. Nat Commun. 2025. PMID: 39753580 Free PMC article.
-
The role of extracellular matrix viscoelasticity in development and disease.NPJ Biol Phys Mech. 2025;2(1):10. doi: 10.1038/s44341-025-00014-6. Epub 2025 Apr 3. NPJ Biol Phys Mech. 2025. PMID: 40191103 Free PMC article. Review.
-
Technological advances and challenges in constructing complex gut organoid systems.Front Cell Dev Biol. 2024 Aug 14;12:1432744. doi: 10.3389/fcell.2024.1432744. eCollection 2024. Front Cell Dev Biol. 2024. PMID: 39206092 Free PMC article. Review.
References
-
- Roda G., Nat. Rev. Dis. Prim. 2020, 6, 22. - PubMed
-
- Kobayashi T., Nat. Rev. Dis. Prim. 2020, 6, 1. - PubMed
-
- Hallberg K., Grzegorczyk A., Larson G., Strandvik B., J. Pediatr. Gastroenterol. Nutr. 1997, 25, 290. - PubMed
-
- Soler A. P., Miller R. D., Laughlin K. V., Carp N. Z., Klurfeld D. M., Mullin J. M., Carcinogenesis 1999, 20, 1425. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
