Origin and development of primary animal epithelia
- PMID: 38009581
- PMCID: PMC11164562
- DOI: 10.1002/bies.202300150
Origin and development of primary animal epithelia
Abstract
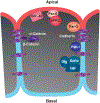
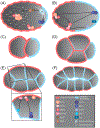
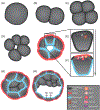
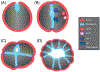
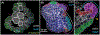
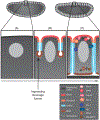
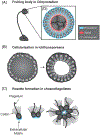
Epithelia are the first organized tissues that appear during development. In many animal embryos, early divisions give rise to a polarized monolayer, the primary epithelium, rather than a random aggregate of cells. Here, we review the mechanisms by which cells organize into primary epithelia in various developmental contexts. We discuss how cells acquire polarity while undergoing early divisions. We describe cases where oriented divisions constrain cell arrangement to monolayers including organization on top of yolk surfaces. We finally discuss how epithelia emerge in embryos from animals that branched early during evolution and provide examples of epithelia-like arrangements encountered in single-celled eukaryotes. Although divergent and context-dependent mechanisms give rise to primary epithelia, here we trace the unifying principles underlying their formation.
Keywords: blastocoel; epithelialization; lumen; monolayer; polarity; self-organization.
© 2023 Wiley Periodicals LLC.
Conflict of interest statement
CONFLICT OF INTEREST STATEMENT
The authors declare no conflicts of interest.
Figures



References
-
- Brusca GJ, Brusca RC, & Gilbert SF (1997). Characteristics of metazoan development. In: (Gilbert AM, S.F. and Raunio Ed.), Embryology: Constructing the Organism (pp. 3–19).
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

