Rising pharmaceutical innovation in the Global South: a landscape study
- PMID: 38012700
- PMCID: PMC10680326
- DOI: 10.1186/s40545-023-00669-3
Rising pharmaceutical innovation in the Global South: a landscape study
Abstract
Background: There is growing interest in pharmaceutical innovation in low- and middle-income countries (LMICs), but information on existing activities, capacities, and outcomes is scarce. We mapped available data at the global level, and studied the national pharmaceutical innovation systems of Bangladesh and Colombia to shed light on pharmaceutical research and development (R&D) in the Global South, including challenges and prospects, to help fill existing knowledge gaps.
Methods: We gathered and analyzed data from three types of sources: literature, semi-structured interviews with key informants, and publicly available data on R&D funding, R&D scientific capacity measured by human resources, and clinical trial activities.
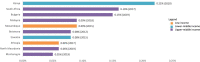
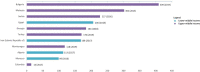
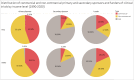
Results: Pharmaceutical R&D activities are occurring in many LMICs, but 16 countries have emerged as frontrunners. Investment in R&D in LMICs has increased in the past decade, particularly from middle-income countries (MICs). Capacity is also growing, with an increase in the number of research organizations and the amount of funding available from external sources. The total number of clinical trials and the proportion of trials in LMICs increased markedly, and there is also growing activity in the earlier, more innovative and riskier Phase 1 and 2 trials. Non-commercial entities comprise the majority of clinical trial funders and sponsors in LMICs. Finally, investments have borne fruit, as indicated by a number of innovative medicines developed in LMICs. The Bangladesh and Colombia country studies showed that there is still a need for both targeted R&D policies to strengthen capacities in the pharmaceutical sector, and more government support to overcome the challenges of a lack of funding and coordination among different actors.
Conclusions: By triangulating between the data sources, it was possible to paint a broad picture of who was involved in pharmaceutical R&D in LMICs, in which particular countries, for which diseases, in which R&D phases, and with what results-as well as how these trends have changed over time. Prioritizing pharmaceutical R&D is an important strategy for better meeting health needs. The trendlines are promising, but focused attention is still needed to realize the potential for greater innovation in the Global South.
Keywords: Access to medicines; Global South; Pharmaceutical innovation; Research and development.
© 2023. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures





References
-
- Akkari ACS, Munhoz IP, Tomioka J, Santos NMBF, Santos RFD. Pharmaceutical innovation: differences between Europe, USA and ‘pharmerging’ Countries. Gest Prod. 2016;23:365–380. doi: 10.1590/0104-530X2150-15. - DOI
-
- Andia T, Alfonso Pineda S, Gomez JS, Vargas MG, Castro C. Colombian health innovation landscape: Building Bridges. Universidad de los Andes; 2023.
-
- Bower DJ, Sulej JC. The Indian challenge: the evolution of a successful new global strategy in the pharmaceutical industry. Technol Anal Strateg Manag. 2007;19(5):611–624. doi: 10.1080/09537320701521358. - DOI
-
- Centro de pensamiento Medicamentos, información y poder. Innovación con enfoque en salud pública - Informe final y portafolio de casos. 2019. http://pensamiento.unal.edu.co/fileadmin/recursos/focos/medicamentos/doc.... Accessed 18 May 2023.
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
