Pharmacokinetics and Biodistribution of 16,16 dimethyl Prostaglandin E2 in Non-Irradiated and Irradiated Mice and Non-Irradiated Non-Human Primates
- PMID: 38019093
- PMCID: PMC11163368
- DOI: 10.1667/RADE-23-00040.1
Pharmacokinetics and Biodistribution of 16,16 dimethyl Prostaglandin E2 in Non-Irradiated and Irradiated Mice and Non-Irradiated Non-Human Primates
Abstract
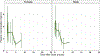
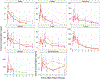
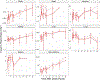
Exposure to high-dose ionizing radiation can lead to life-threatening injuries and mortality. Bone marrow is the most sensitive organ to radiation damage, resulting in the hematopoietic acute radiation syndrome (H-ARS) with the potential sequelae of infection, hemorrhage, anemia, and death if untreated. The development of medical countermeasures (MCMs) to protect or mitigate radiation injury is a medical necessity. In our well-established murine model of H-ARS we have demonstrated that the prostaglandin E2 (PGE2) analog 16,16 dimethyl-PGE2 (dmPGE2) has survival efficacy as both a radioprotectant and radiomitigator. The purpose of this study was to investigate the pharmacokinetics (PK) and biodistribution of dmPGE2 when used as a radioprotector in irradiated and non-irradiated inbred C57BL/6J mice, PK in irradiated and non-irradiated Jackson Diversity Outbred (JDO) mice, and the PK profile of dmPGE2 in non-irradiated non-human primates (NHPs). The C57BL/6J and JDO mice each received a single subcutaneous (SC) dose of 35 ug of dmPGE2 and were randomized to either receive radiation 30 min later or remain non-irradiated. Plasma and tissue PK profiles were established. The NHP were dosed with 0.1 mg/kg by SC administration and the PK profile in plasma was established. The concentration time profiles were analyzed by standard non-compartmental analysis and the metrics of AUC0-Inf, AUC60-480 (AUC from 60-480 min), Cmax, and t1/2 were evaluated. AUC60-480 represents the postirradiation time frame and was used to assess radiation effect. Overall, AUC0-Inf, Cmax, and t1/2 were numerically similar between strains (C57BL/6J and JDO) when combined, regardless of exposure status (AUC0-Inf: 112.50 ng·h/ml and 114.48 ng·h/ml, Cmax: 44.53 ng/ml and 63.96 ng/ml; t1/2: 1.8 h and 1.1 h, respectively). PK metrics were numerically lower in irradiated C57BL/6J mice than in non-irradiated mice [irradiation ratio: irradiated values/non-irradiated values = 0.71 for AUC60-480 (i.e., 29% lower), and 0.6 for t1/2]. In JDO mice, the radiation ratio was 0.53 for AUC60-480 (i.e., 47% lower), and 1.7 h for t1/2. The AUC0-Inf, Cmax, and t1/2 of the NHPs were 29.20 ng·h/ml, 7.68 ng/ml, and 3.26 h, respectively. Despite the numerical differences seen between irradiated and non-irradiated groups in PK parameters, the effect of radiation on PK can be considered minimal based on current data. The biodistribution in C57BL/6J mice showed that dmPGE2 per gram of tissue was highest in the lungs, regardless of exposure status. The radiation ratio for the different tissue AUC60-480 in C57BL/6J mice ranged between 0.5-1.1 (50% lower to 10% higher). Spleen, liver and bone marrow showed close to twice lower exposures after irradiation, whereas heart had a 10% higher exposure. Based on the clearance values from mice and NHP, the estimated allometric scaling coefficient was 0.81 (95% CI: 0.75, 0.86). While slightly higher than the current literature estimates of 0.75, this scaling coefficient can be considered a reasonable estimate and can be used to scale dmPGE2 dosing from animals to humans for future trials.
© 2024 by Radiation Research Society.
Figures

References
-
- Till JE, McCulloch EA. Repair Processes in Irradiated Mouse Hematopoietic Tissue. Annals of the New York Academy of Sciences. 1964; 114(1):115–25. - PubMed
-
- Coleman CN, Blakely WF, Fike JR, MacVittie TJ, Metting NF, Mitchell JB, et al. Molecular and cellular biology of moderate-dose (1–10 Gy) radiation and potential mechanisms of radiation protection: report of a workshop at Bethesda, Maryland, December 17–18, 2001. Radiat Res. 2003. Jun; 159(6):812–34. - PubMed
-
- Dainiak N, Waselenko JK, Armitage JO, MacVittie TJ, Farese AM. The hematologist and radiation casualties. Hematology Am Soc Hematol Educ Program. 2003; 473–96. - PubMed
-
- FDA label. Neupogen (filgrastim) [Internet]. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2016/103353s5188.pdf
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

