HPV-associated head and neck cancer is characterized by distinct profiles of CD8+ T cells and myeloid-derived suppressor cells
- PMID: 38019346
- PMCID: PMC10700222
- DOI: 10.1007/s00262-023-03571-8
HPV-associated head and neck cancer is characterized by distinct profiles of CD8+ T cells and myeloid-derived suppressor cells
Abstract
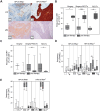
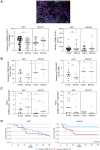
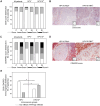
Patients with HPV--localized head and neck cancer (HNC) show inferior outcomes after surgery and radiochemotherapy compared to HPV-associated cancers. The underlying mechanisms remain elusive, but differences in immune status and immune activity may be implicated. In this study, we analyzed immune profiles of CD8+ T cells and myeloid-derived suppressor cells (MDSC) in HPV+ versus HPV- disease.The overall frequency of CD8+ T cells was reduced in HNC versus healthy donors but substantially increased after curative therapy (surgery and/or radiochemotherapy). In HPV+ patients, this increase was associated with significant induction of peripheral blood CD8+/CD45RA-/CD62L- effector memory cells. The frequency of HPV-antigen-specific CD8+ cells was low even in patients with virally associated tumors and dropped to background levels after curative therapy. Pre-therapeutic counts of circulating monocytic MDSC, but not PMN-MDSC, were increased in patients with HPV- disease. This increase was accompanied by reduced fractions of terminally differentiated CD8+ effector cells. HPV- tumors showed reduced infiltrates of CD8+ and CD45RO+ immune cells compared with HPV+ tumors. Importantly, frequencies of tumor tissue-infiltrating PMN-MDSC were increased, while percentages of Granzyme B+ and Ki-67+ CD8 T cells were reduced in patients with HPV- disease.We report differences in frequencies and relative ratios of MDSC and effector T cells in HPV- HNC compared with more immunogenic HPV-associated disease. Our data provide new insight into the immunological profiles of these two tumor entities and may be utilized for more tailored immunotherapeutic approaches in the future.
Keywords: CD8+ T cells; HPV; Head and neck cancer; MDSC; Oropharyngeal cancer.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no financial interests. Sven Brandau is a member of the journals’ editorial board.
Figures




Similar articles
-
Protection against HPV-16-Associated Tumors Requires the Activation of CD8+ Effector Memory T Cells and the Control of Myeloid-Derived Suppressor Cells.Mol Cancer Ther. 2016 Aug;15(8):1920-30. doi: 10.1158/1535-7163.MCT-15-0742. Epub 2016 May 24. Mol Cancer Ther. 2016. PMID: 27222537
-
Clinical Relevance and Suppressive Capacity of Human Myeloid-Derived Suppressor Cell Subsets.Clin Cancer Res. 2018 Oct 1;24(19):4834-4844. doi: 10.1158/1078-0432.CCR-17-3726. Epub 2018 Jun 18. Clin Cancer Res. 2018. PMID: 29914893
-
Effector CD8+CD45RO-CD27-T cells have signalling defects in patients with squamous cell carcinoma of the head and neck.Br J Cancer. 2003 Jan 27;88(2):223-30. doi: 10.1038/sj.bjc.6600694. Br J Cancer. 2003. PMID: 12610507 Free PMC article.
-
Impact of HPV Infection on the Immune System in Oropharyngeal and Non-Oropharyngeal Squamous Cell Carcinoma: A Systematic Review.Cells. 2019 Sep 10;8(9):1061. doi: 10.3390/cells8091061. Cells. 2019. PMID: 31510065 Free PMC article.
-
[Comparison of T cell response in the tumor milieu of patients with HPV+ and HPV- head and neck cancer].HNO. 2020 Feb;68(2):80-86. doi: 10.1007/s00106-019-00804-0. HNO. 2020. PMID: 31915881 Review. German.
Cited by
-
The responses of HNSCC patients to immunotherapy are shown by two novel co-expression patterns.NPJ Precis Oncol. 2025 Jul 1;9(1):217. doi: 10.1038/s41698-025-00983-w. NPJ Precis Oncol. 2025. PMID: 40596712 Free PMC article.
-
Immune Evasion in Head and Neck Squamous Cell Carcinoma: Roles of Cancer-Associated Fibroblasts, Immune Checkpoints, and TP53 Mutations in the Tumor Microenvironment.Cancers (Basel). 2025 Aug 7;17(15):2590. doi: 10.3390/cancers17152590. Cancers (Basel). 2025. PMID: 40805285 Free PMC article. Review.
-
Type I conventional dendritic cells and CD8+ T cells predict favorable clinical outcome of head and neck squamous cell carcinoma patients.Front Immunol. 2024 Jun 13;15:1414298. doi: 10.3389/fimmu.2024.1414298. eCollection 2024. Front Immunol. 2024. PMID: 38938577 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

