Chaos in a bacterial stress response
- PMID: 38029757
- PMCID: PMC7616676
- DOI: 10.1016/j.cub.2023.11.002
Chaos in a bacterial stress response
Abstract
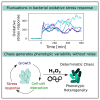
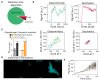
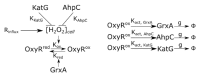
Cellular responses to environmental changes are often highly heterogeneous and exhibit seemingly random dynamics. The astonishing insight of chaos theory is that such unpredictable patterns can, in principle, arise without the need for any random processes, i.e., purely deterministically without noise. However, while chaos is well understood in mathematics and physics, its role in cell biology remains unclear because the complexity and noisiness of biological systems make testing difficult. Here, we show that chaos explains the heterogeneous response of Escherichia coli cells to oxidative stress. We developed a theoretical model of the gene expression dynamics and demonstrate that chaotic behavior arises from rapid molecular feedbacks that are coupled with cell growth dynamics and cell-cell interactions. Based on theoretical predictions, we then designed single-cell experiments to show we can shift gene expression from periodic oscillations to chaos on demand. Our work suggests that chaotic gene regulation can be employed by cell populations to generate strong and variable responses to changing environments.
Keywords: bacterial stress response; chaos; gene regulation; oxidative stress; phenotypic heterogeneity; single-cell analysis.
Copyright © 2023 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no competing interests.
Figures





References
-
- Strogatz SH. Nonlinear Dynamics and Chaos: With Applications to Physics, Biology, Chemistry, and Engineering. Westview Press; 2018.
-
- Rogers TL, Johnson BJ, Munch SB. Chaos is not rare in natural ecosystems. Nat Ecol Evol. 2022;6:1105–1111. - PubMed
-
- Allen JC, Schaffer WM, Rosko D. Chaos reduces species extinction by amplifying local population noise. Nature. 1993;364:229–232. - PubMed
-
- Cenci S, Saavedra S. Non-parametric estimation of the structural stability of non-equilibrium community dynamics. Nat Ecol Evol. 2019;3:912–918. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

