EpCAM-targeting CAR-T cell immunotherapy is safe and efficacious for epithelial tumors
- PMID: 38039357
- PMCID: PMC10691766
- DOI: 10.1126/sciadv.adg9721
EpCAM-targeting CAR-T cell immunotherapy is safe and efficacious for epithelial tumors
Abstract
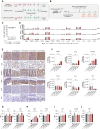
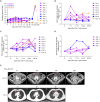
The efficacy of CAR-T cells for solid tumors is unsatisfactory. EpCAM is a biomarker of epithelial tumors, but the clinical feasibility of CAR-T therapy targeting EpCAM is lacking. Here, we report pre- and clinical investigations of EpCAM-CAR-T cells for solid tumors. We demonstrated that EpCAM-CAR-T cells costimulated by Dectin-1 exhibited robust antitumor activity without adverse effects in xenograft mouse models and EpCAM-humanized mice. Notably, in clinical trials for epithelial tumors (NCT02915445), 6 (50%) of the 12 enrolled patients experienced self-remitted grade 1/2 toxicities, 1 patient (8.3%) experienced reversible grade 3 leukopenia, and no higher-grade toxicity reported. Efficacy analysis determined two patients as partial response. Three patients showed >23 months of progression-free survival, among whom one patient experienced 2-year progress-free survival with detectable CAR-T cells 200 days after infusion. These data demonstrate the feasibility and tolerability of EpCAM-CAR-T therapy.
Figures



References
-
- S. J. Schuster, M. R. Bishop, C. S. Tam, E. K. Waller, P. Borchmann, J. P. McGuirk, U. Jäger, S. Jaglowski, C. Andreadis, J. R. Westin, I. Fleury, V. Bachanova, S. R. Foley, P. J. Ho, S. Mielke, J. M. Magenau, H. Holte, S. Pantano, L. B. Pacaud, R. Awasthi, J. Chu, Ö. Anak, G. Salles, R. T. Maziarz; JULIET Investigators , Tisagenlecleucel in adult relapsed or refractory diffuse large B-cell lymphoma. N. Engl. J. Med. 380, 45–56 (2019). - PubMed
-
- E. A. Chong, M. Ruella, S. J. Schuster; Lymphoma Program Investigators at the University of Pennsylvania , Five-year outcomes for refractory B-cell lymphomas with CAR T-cell therapy. N. Engl. J. Med. 384, 673–674 (2021). - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

