COVID-19 Boosters: If The US Had Matched Israel's Speed And Take-Up, An Estimated 29,000 US Lives Would Have Been Saved
- PMID: 38048511
- PMCID: PMC11470653
- DOI: 10.1377/hlthaff.2023.00718
COVID-19 Boosters: If The US Had Matched Israel's Speed And Take-Up, An Estimated 29,000 US Lives Would Have Been Saved
Abstract
Israel was the first country to launch COVID-19 boosters, in late July 2021, with strong public health messaging. The booster campaign reversed rising infection rates from the Delta variant and reduced hospitalizations and deaths. The US booster rollout was slower, and public health messaging was mixed. We used the Israeli experience to ask the counterfactual question: How many lives could the US have saved if it had authorized boosters sooner? We estimated that through June 30, 2022, if the US had moved at Israel's speed and booster take-up percentages, it would have saved 29,000 lives. US regulatory caution, in the middle of a pandemic, thus had a large, avoidable cost. Yet the US booster rollout still avoided 42,000 deaths. Moving more slowly to approve boosters, as some advocated, would have cost many additional lives.
Figures


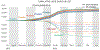
Comment on
-
Considerations in boosting COVID-19 vaccine immune responses.Lancet. 2021 Oct 9;398(10308):1377-1380. doi: 10.1016/S0140-6736(21)02046-8. Epub 2021 Sep 14. Lancet. 2021. PMID: 34534516 Free PMC article. Review. No abstract available.
References
-
- To access the appendix, click on the Details tab of the article online.
-
- Neergaard L. Looking at delta variant, Pfizer to seek FDA authorization for 3rd vaccine dose. Times of Israel [serial on the Internet]. 2021. Jul 9 [cited 2023 Nov 7]. Available from: https://www.timesofisrael.com/looking-at-delta-variant-pfizer-to-seek-fd...
-
- Centers for Disease Control and Prevention [Internet]. Atlanta (GA): CDC. Press release, Joint statement from HHS public health and medical experts on COVID-19 booster shots; 2021. Aug 18 [cited 2023 Oct 16]. Available from: https://www.cdc.gov/media/releases/2021/s0818-covid-19-booster-shots.html
Publication types
MeSH terms
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

