NK92 Expressing Anti-BCMA CAR and Secreted TRAIL for the Treatment of Multiple Myeloma: Preliminary In Vitro Assessment
- PMID: 38067177
- PMCID: PMC10706019
- DOI: 10.3390/cells12232748
NK92 Expressing Anti-BCMA CAR and Secreted TRAIL for the Treatment of Multiple Myeloma: Preliminary In Vitro Assessment
Abstract
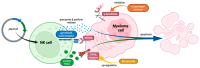
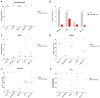
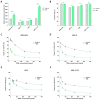
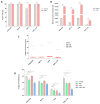
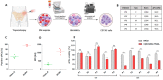
Multiple myeloma (MM) has witnessed improved patient outcomes through advancements in therapeutic approaches. Notably, allogeneic stem cell transplantation, proteasome inhibitors, immunomodulatory drugs, and monoclonal antibodies have contributed to enhanced quality of life. Recently, a promising avenue has emerged with chimeric antigen receptor (CAR) T cells targeting B-cell maturation antigen (BCMA), expressed widely on MM cells. To mitigate risks associated with allogenic T cells, we investigated the potential of BCMA CAR expression in natural killer cells (NKs), known for potent cytotoxicity and minimal side effects. Using the NK-92 cell line, we co-expressed BCMA CAR and soluble tumor necrosis factor-related apoptosis-inducing ligand (sTRAIL) employing the piggyBac transposon system. Engineered NK cells (CAR-NK-92-TRAIL) demonstrated robust cytotoxicity against a panel of MM cell lines and primary patient samples, outperforming unmodified NK-92 cells with a mean difference in viability of 45.1% (±26.1%, depending on the target cell line). Combination therapy was explored with the proteasome inhibitor bortezomib (BZ) and γ-secretase inhibitors (GSIs), leading to a significant synergistic effect in combination with CAR-NK-92-TRAIL cells. This synergy was evident in cytotoxicity assays where a notable decrease in MM cell viability was observed in combinatorial therapy compared to single treatment. In summary, our study demonstrates the therapeutic potential of the CAR-NK-92-TRAIL cells for the treatment of MM. The synergistic impact of combining these engineered NK cells with BZ and GSI supports further development of allogeneic CAR-based products for effective MM therapy.
Keywords: allogenic; cancer; chimeric antigen receptor; immunotherapy; multiple myeloma; natural killer.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Bortezomib enhances the efficacy of BCMA CAR-T therapy through up-regulating BCMA expression in myeloma cells.Int Immunopharmacol. 2025 Feb 20;148:114113. doi: 10.1016/j.intimp.2025.114113. Epub 2025 Jan 24. Int Immunopharmacol. 2025. PMID: 39862629
-
CXCR4 has a dual role in improving the efficacy of BCMA-redirected CAR-NK cells in multiple myeloma.Front Immunol. 2024 Jun 24;15:1383136. doi: 10.3389/fimmu.2024.1383136. eCollection 2024. Front Immunol. 2024. PMID: 38979422 Free PMC article.
-
γ-Secretase inhibition increases efficacy of BCMA-specific chimeric antigen receptor T cells in multiple myeloma.Blood. 2019 Nov 7;134(19):1585-1597. doi: 10.1182/blood.2019000050. Blood. 2019. PMID: 31558469 Free PMC article.
-
Targeting B Cell Maturation Antigen (BCMA) in Multiple Myeloma: Potential Uses of BCMA-Based Immunotherapy.Front Immunol. 2018 Aug 10;9:1821. doi: 10.3389/fimmu.2018.01821. eCollection 2018. Front Immunol. 2018. PMID: 30147690 Free PMC article. Review.
-
Recent updates on CAR T clinical trials for multiple myeloma.Mol Cancer. 2019 Nov 5;18(1):154. doi: 10.1186/s12943-019-1092-1. Mol Cancer. 2019. PMID: 31684964 Free PMC article. Review.
Cited by
-
Engineered exosomes in emerging cell-free therapy.Front Oncol. 2024 Mar 26;14:1382398. doi: 10.3389/fonc.2024.1382398. eCollection 2024. Front Oncol. 2024. PMID: 38595822 Free PMC article. Review.
-
Rendering NK Cells Antigen-Specific for the Therapy of Solid Tumours.Int J Mol Sci. 2025 Jun 29;26(13):6290. doi: 10.3390/ijms26136290. Int J Mol Sci. 2025. PMID: 40650066 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

