Mutation Status and Glucose Availability Affect the Response to Mitochondria-Targeted Quercetin Derivative in Breast Cancer Cells
- PMID: 38067318
- PMCID: PMC10705313
- DOI: 10.3390/cancers15235614
Mutation Status and Glucose Availability Affect the Response to Mitochondria-Targeted Quercetin Derivative in Breast Cancer Cells
Abstract
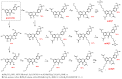
Mitochondria, the main cellular power stations, are important modulators of redox-sensitive signaling pathways that may determine cell survival and cell death decisions. As mitochondrial function is essential for tumorigenesis and cancer progression, mitochondrial targeting has been proposed as an attractive anticancer strategy. In the present study, three mitochondria-targeted quercetin derivatives (mitQ3, 5, and 7) were synthesized and tested against six breast cancer cell lines with different mutation and receptor status, namely ER-positive MCF-7, HER2-positive SK-BR-3, and four triple-negative (TNBC) cells, i.e., MDA-MB-231, MDA-MB-468, BT-20, and Hs 578T cells. In general, the mito-quercetin response was modulated by the mutation status. In contrast to unmodified quercetin, 1 µM mitQ7 induced apoptosis in breast cancer cells. In MCF-7 cells, mitQ7-mediated apoptosis was potentiated under glucose-depleted conditions and was accompanied by elevated mitochondrial superoxide production, while AMPK activation-based energetic stress was associated with the alkalization of intracellular milieu and increased levels of NSUN4. Mito-quercetin also eliminated doxorubicin-induced senescent breast cancer cells, which was accompanied by the depolarization of mitochondrial transmembrane potential. Limited glucose availability also sensitized doxorubicin-induced senescent breast cancer cells to apoptosis. In conclusion, we show an increased cytotoxicity of mitochondria-targeted quercetin derivatives compared to unmodified quercetin against breast cancer cells with different mutation status that can be potentiated by modulating glucose availability.
Keywords: AMPK; breast cancer; doxorubicin-induced senescence; mito-quercetin; oxidative stress; senolysis.
Conflict of interest statement
The authors have no conflict of interest to disclose.
Figures



Similar articles
-
New Mitochondria-Targeted Fisetin Derivative Compromises Mitophagy and Limits Survival of Drug-Induced Senescent Breast Cancer Cells.J Med Chem. 2024 Oct 10;67(19):17676-17689. doi: 10.1021/acs.jmedchem.4c01664. Epub 2024 Sep 25. J Med Chem. 2024. PMID: 39322603 Free PMC article.
-
Mitochondrial transplantation regulates antitumour activity, chemoresistance and mitochondrial dynamics in breast cancer.J Exp Clin Cancer Res. 2019 Jan 23;38(1):30. doi: 10.1186/s13046-019-1028-z. J Exp Clin Cancer Res. 2019. PMID: 30674338 Free PMC article.
-
Lapatinib- and fulvestrant-PAMAM dendrimer conjugates promote apoptosis in chemotherapy-induced senescent breast cancer cells with different receptor status.Biomater Adv. 2022 Sep;140:213047. doi: 10.1016/j.bioadv.2022.213047. Epub 2022 Jul 26. Biomater Adv. 2022. PMID: 35917687
-
Senolysis-Based Elimination of Chemotherapy-Induced Senescent Breast Cancer Cells by Quercetin Derivative with Blocked Hydroxy Groups.Cancers (Basel). 2022 Jan 25;14(3):605. doi: 10.3390/cancers14030605. Cancers (Basel). 2022. PMID: 35158873 Free PMC article.
-
The novel camptothecin derivative, CPT211, induces cell cycle arrest and apoptosis in models of human breast cancer.Biomed Pharmacother. 2020 Aug;128:110309. doi: 10.1016/j.biopha.2020.110309. Epub 2020 Jun 4. Biomed Pharmacother. 2020. PMID: 32505820
Cited by
-
Quercetin in Oncology: A Phytochemical with Immense Therapeutic Potential.Curr Drug Targets. 2024;25(11):740-751. doi: 10.2174/0113894501292466240627050638. Curr Drug Targets. 2024. PMID: 38988154 Review.
-
Quercetin Promote the Chemosensitivity in Organoids Derived from Patients with Breast Cancer.Breast Cancer (Dove Med Press). 2024 Dec 20;16:993-1004. doi: 10.2147/BCTT.S494901. eCollection 2024. Breast Cancer (Dove Med Press). 2024. PMID: 39720358 Free PMC article.
-
Anticancer Activity of Encapsulated Pearl Millet Polyphenol-Rich Extract against Proliferating and Non-Proliferating Breast Cancer Cells In Vitro.Cancers (Basel). 2024 Apr 30;16(9):1750. doi: 10.3390/cancers16091750. Cancers (Basel). 2024. PMID: 38730703 Free PMC article.
-
Quercetin: A Natural Ally in Combating Breast Cancer.Int J Nanomedicine. 2025 Jul 19;20:9155-9177. doi: 10.2147/IJN.S518174. eCollection 2025. Int J Nanomedicine. 2025. PMID: 40708864 Free PMC article. Review.
-
New Mitochondria-Targeted Fisetin Derivative Compromises Mitophagy and Limits Survival of Drug-Induced Senescent Breast Cancer Cells.J Med Chem. 2024 Oct 10;67(19):17676-17689. doi: 10.1021/acs.jmedchem.4c01664. Epub 2024 Sep 25. J Med Chem. 2024. PMID: 39322603 Free PMC article.
References
-
- Tan A.S., Baty J.W., Dong L.-F., Bezawork-Geleta A., Endaya B., Goodwin J., Bajzikova M., Kovarova J., Peterka M., Yan B., et al. Mitochondrial Genome Acquisition Restores Respiratory Function and Tumorigenic Potential of Cancer Cells without Mitochondrial DNA. Cell Metab. 2015;21:81–94. doi: 10.1016/j.cmet.2014.12.003. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

