Clonal Neoantigen: Emerging "Mechanism-based" Biomarker of Immunotherapy Response
- PMID: 38067319
- PMCID: PMC10705148
- DOI: 10.3390/cancers15235616
Clonal Neoantigen: Emerging "Mechanism-based" Biomarker of Immunotherapy Response
Abstract
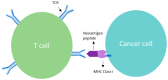
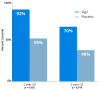
Clonal mutations represent the initiating molecular defects related to cellular transition of a normal phenotype to a malignant phenotype. Molecular genomic assessment utilizing next generation and whole exome sequencing is now being increasingly applied to biomarker determination to refine the use of targeted immune therapies. Case examples followed by retrospective study assessment have convincingly demonstrated clonal neoantigens provide a relevant predictor of response to checkpoint inhibition. A meta-analysis, by Litchfield et al., of over 1000 cancer patients from 12 landmark trials demonstrated no clinical benefit to checkpoint inhibitor (CPI) therapy in correlation to high subclonal tumor mutational burden (TMB), whereas high clonal TMB was found to be significantly correlated with better overall survival (p = 0.000000029). We discuss the mechanism of clonal vs. subclonal neoantigen targeting relationship to homologous recombination proficient (HRP) profile, evidence of preclinical and clinical benefit related to clonal neoantigens, and review a novel developing therapy called Vigil®, designed to expand the clonal neoantigen targeting effector cell populations. Vigil® is an autologous cellular immunotherapy which is designed to carry the full set of personal clonal neoantigens. Phase 2b results demonstrate a durable recurrence-free survival (RFS) and overall survival (OS) advantage for Vigil® in a subset ovarian cancer population with an HRP cancer profile.
Keywords: Vigil; biomarker; cancer; clonal neoantigen; immunotherapy.
Conflict of interest statement
J.N reports employment, stockholder and Board member of Gradalis, Inc. L.S. and A.W. are employees of Gradalis, Inc. D.W. is an employee of Frontage Laboratories, Inc. E.B. reports stockholder and employee of Gradalis, Inc. S.B. reports being a paid consultant for Gradalis, Inc. B.J.M. reports being a paid consultant for Acrivon, Adaptimmune, Agenus, Akeso Bio, Amgen, Aravive, AstraZeneca, Bayer, Clovis, Easai, Elevar, EMD Merck, Genmab/Seagen, GOG Foundation, Gradalis, Inc., Heng Rui, ImmunoGen, Karyopharm, Iovance, Laekna Health Care, Merck, Mersana, Myriad, Novartis, Novocure, OncoC4, Panavance, Pieris, Pfizer, Puma, Regeneron, Roche/Genentech, Sorrento, TESARO/GSK, US Oncology Research, VBL, Verastem, Zentalis. R.P.R. reports being paid consultant for Gradalis, Inc., ImmunoGen, Easai, and GSK. KC has no conflict of interest to declare. R.L.C. reports grants and contracts from AstraZeneca, Clovis, Genelux, Genmab, Merck, Immunogen, Roche/Genentech; paid consultant for Agenus, Alkermes, AstraZeneca, Clovis, Deciphera, Genelux, Genmab, GSK, Immunogen, OncoQuest, Onxerna, Regeneron, Roche/Genentech, Novocure, Merck, Abbvie; participation on a data safety monitoring board or advisory board for NRG Oncology and Eisai/BMS.
Figures


Similar articles
-
Gemogenovatucel-T (Vigil): bi-shRNA plasmid-based targeted immunotherapy.Future Oncol. 2024;20(29):2149-2164. doi: 10.1080/14796694.2024.2376518. Epub 2024 Aug 5. Future Oncol. 2024. PMID: 39101448 Free PMC article. Review.
-
Gemogenovatucel-T (Vigil) maintenance immunotherapy: 3-year survival benefit in homologous recombination proficient (HRP) ovarian cancer.Gynecol Oncol. 2021 Dec;163(3):459-464. doi: 10.1016/j.ygyno.2021.10.004. Epub 2021 Oct 23. Gynecol Oncol. 2021. PMID: 34702567
-
Gemogenovatucel-T (Vigil) immunotherapy demonstrates clinical benefit in homologous recombination proficient (HRP) ovarian cancer.Gynecol Oncol. 2021 Jun;161(3):676-680. doi: 10.1016/j.ygyno.2021.03.009. Epub 2021 Mar 11. Gynecol Oncol. 2021. PMID: 33715892 Clinical Trial.
-
Network based analysis identifies TP53m-BRCA1/2wt-homologous recombination proficient (HRP) population with enhanced susceptibility to Vigil immunotherapy.Cancer Gene Ther. 2022 Jul;29(7):993-1000. doi: 10.1038/s41417-021-00400-x. Epub 2021 Nov 16. Cancer Gene Ther. 2022. PMID: 34785763 Free PMC article. Clinical Trial.
-
Cancer neoantigens as potential targets for immunotherapy.Clin Exp Metastasis. 2022 Feb;39(1):51-60. doi: 10.1007/s10585-021-10091-1. Epub 2021 May 5. Clin Exp Metastasis. 2022. PMID: 33950415 Free PMC article. Review.
Cited by
-
Gemogenovatucel-T (Vigil): bi-shRNA plasmid-based targeted immunotherapy.Future Oncol. 2024;20(29):2149-2164. doi: 10.1080/14796694.2024.2376518. Epub 2024 Aug 5. Future Oncol. 2024. PMID: 39101448 Free PMC article. Review.
-
Exome sequencing shows same pattern of clonal tumor mutational burden, intratumor heterogenicity and clonal neoantigen between autologous tumor and Vigil product.Sci Rep. 2025 Mar 13;15(1):8637. doi: 10.1038/s41598-025-90136-7. Sci Rep. 2025. PMID: 40082566 Free PMC article. Clinical Trial.
-
Neoantigen load as a predictor of relapse in early-stage NSCLC: features that agonise and antagonise prognosis.Cancer Immunol Immunother. 2025 Aug 6;74(9):285. doi: 10.1007/s00262-025-04131-y. Cancer Immunol Immunother. 2025. PMID: 40770517 Free PMC article.
-
Clinical characterization and therapeutic targeting of fusion genes in oncology.Future Oncol. 2025 Apr;21(10):1249-1260. doi: 10.1080/14796694.2025.2477974. Epub 2025 Mar 24. Future Oncol. 2025. PMID: 40128124 Free PMC article. Review.
-
Exploring the causal relationship between immune factors and chondrosarcoma: a Mendelian randomization study.Discov Oncol. 2025 May 18;16(1):801. doi: 10.1007/s12672-025-02654-5. Discov Oncol. 2025. PMID: 40382743 Free PMC article.
References
-
- Wilmott J.S., Tembe V., Howle J.R., Sharma R., Thompson J.F., Rizos H., Lo R.S., Kefford R.F., Scolyer R.A., Long G.V. Intratumoral molecular heterogeneity in a BRAF-mutant, BRAF inhibitor-resistant melanoma: A case illustrating the challenges for personalized medicine. Mol. Cancer Ther. 2012;11:2704–2708. doi: 10.1158/1535-7163.MCT-12-0530. - DOI - PubMed
-
- Chen Z.Y., Zhong W.Z., Zhang X.C., Su J., Yang X.N., Chen Z.H., Yang J.J., Zhou Q., Yan H.H., An S.J., et al. EGFR mutation heterogeneity and the mixed response to EGFR tyrosine kinase inhibitors of lung adenocarcinomas. Oncologist. 2012;17:978–985. doi: 10.1634/theoncologist.2011-0385. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

