The Role of ZNF275/AKT Pathway in Carcinogenesis and Cisplatin Chemosensitivity of Cervical Cancer Using Patient-Derived Xenograft Models
- PMID: 38067329
- PMCID: PMC10705782
- DOI: 10.3390/cancers15235625
The Role of ZNF275/AKT Pathway in Carcinogenesis and Cisplatin Chemosensitivity of Cervical Cancer Using Patient-Derived Xenograft Models
Abstract
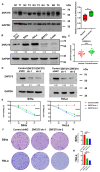
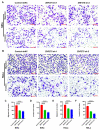
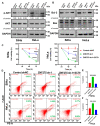
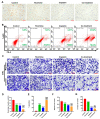
Zinc finger protein 275 (ZNF275) is a C2H2-type transcription factor that is localized on chromosome Xq28. Whether ZNF275 participates in modulating the biological behaviors of cervical cancer has not been determined to our knowledge. The present study employed CCK-8, BrdU, flow cytometry, and a transwell assay to investigate the cell viability, proliferation, apoptosis, migration, and invasion of cervical cancer cells. The application of Western blotting and immunohistochemistry (IHC) aims to assess ZNF275 protein expression and identify the signaling pathway relevant to ZNF275-mediated effects on cervical cancer. The therapeutic impact of the combined therapy of the AKT inhibitor triciribine and cisplatin was evaluated on cervical cancer patient-derived xenograft (PDX) models expressing high ZNF275. The current research illustrated that cervical cancer tissue exhibited a higher expression of ZNF275 in contrast to the surrounding normal cervical tissue. The downregulation of ZNF275 suppressed cell viability, migration, and invasion, and facilitated the apoptosis of SiHa and HeLa cells via weakening AKT/Bcl-2 signaling pathway. Moreover, triciribine synergized with cisplatin to reduce cell proliferation, migration, and invasion, and enhanced the apoptosis of SiHa cells expressing high ZNF275. In addition, the combination treatment of triciribine and cisplatin was more effective in inducing tumor regression than single agents in cervical cancer PDX models expressing high ZNF275. Collectively, the current findings demonstrated that ZNF275 serves as a sufficiently predictive indicator of the therapeutic effectiveness of the combined treatment of triciribine and cisplatin on cervical cancer. Combining triciribine with cisplatin greatly broadens the therapeutic options for cervical cancer expressing high ZNF275, but further research is needed to confirm these results.
Keywords: cervical cancer; chemosensitivity; cisplatin; patient-derived xenograft (PDX) models; zinc finger protein 275 (ZNF275).
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
TMEM45A Affects Proliferation, Apoptosis, Epithelial-Mesenchymal Transition, Migration, Invasion and Cisplatin Resistance of HPV-Positive Cervical Cancer Cell Lines.Biochem Genet. 2022 Feb;60(1):173-190. doi: 10.1007/s10528-021-10094-3. Epub 2021 Jun 18. Biochem Genet. 2022. PMID: 34143331
-
The Antitumor Efficiency of Zinc Finger Nuclease Combined with Cisplatin and Trichostatin A in Cervical Cancer Cells.Anticancer Agents Med Chem. 2020;20(17):2125-2135. doi: 10.2174/1871520620666200804102300. Anticancer Agents Med Chem. 2020. PMID: 32753022
-
PSAT1 Upregulation Contributes to Cell Growth and Cisplatin Resistance in Cervical Cancer Cells via Regulating PI3K/AKT Signaling Pathway.Ann Clin Lab Sci. 2020 Jul;50(4):512-518. Ann Clin Lab Sci. 2020. PMID: 32826249
-
Effects of Quercetin on the Efficacy of Various Chemotherapeutic Drugs in Cervical Cancer Cells.Drug Des Devel Ther. 2021 Feb 15;15:577-588. doi: 10.2147/DDDT.S291865. eCollection 2021. Drug Des Devel Ther. 2021. PMID: 33623367 Free PMC article.
-
Cisplatin inhibits the growth, migration and invasion of cervical cancer cells by down-regulating IL-17E/IL-17RB.Int J Clin Exp Pathol. 2017 Sep 1;10(9):9341-9351. eCollection 2017. Int J Clin Exp Pathol. 2017. PMID: 31966806 Free PMC article.
Cited by
-
Patient-derived xenograft models: Current status, challenges, and innovations in cancer research.Genes Dis. 2025 Jan 8;12(5):101520. doi: 10.1016/j.gendis.2025.101520. eCollection 2025 Sep. Genes Dis. 2025. PMID: 40548062 Free PMC article. Review.
-
Dual Topoisomerase Inhibitor Is Highly Potent and Improves Antitumor Response to Radiotherapy in Cervical Carcinoma.Int J Mol Sci. 2025 Mar 21;26(7):2829. doi: 10.3390/ijms26072829. Int J Mol Sci. 2025. PMID: 40243435 Free PMC article.
-
Cancer Chemotherapy: Combination with Inhibitors (Volume I).Cancers (Basel). 2024 Jan 31;16(3):607. doi: 10.3390/cancers16030607. Cancers (Basel). 2024. PMID: 38339356 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources

