Antidiabetic Activity, Molecular Docking, and ADMET Properties of Compounds Isolated from Bioactive Ethyl Acetate Fraction of Ficus lutea Leaf Extract
- PMID: 38067448
- PMCID: PMC10708125
- DOI: 10.3390/molecules28237717
Antidiabetic Activity, Molecular Docking, and ADMET Properties of Compounds Isolated from Bioactive Ethyl Acetate Fraction of Ficus lutea Leaf Extract
Abstract
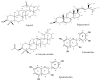
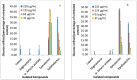
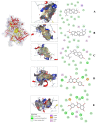
Diabetes contributes to the rising global death rate. Despite scientific advancements in understanding and managing diabetes, no single therapeutic agent has been identified to effectively treat and prevent its progression. Consequently, the exploration for new antidiabetic therapeutics continues. This study aimed to investigate the antidiabetic bioactive ethyl acetate fraction of F. lutea at the molecular level to understand the molecular interactions and ligand-protein binding. To do this, the fraction underwent column chromatography fractionation to yield five compounds: lupeol, stigmasterol, α-amyrin acetate, epicatechin, and epiafzelechin. These compounds were evaluated in vitro through α-glucosidase inhibition and glucose utilization assays in C2C12 muscle and H-4-11-E liver cells using standard methods. In silico analysis was conducted using molecular docking and ADMET studies. Epicatechin exhibited the most potent α-glucosidase inhibition (IC50 = 5.72 ± 2.7 µg/mL), while epiafzelechin stimulated superior glucose utilization in C2C12 muscle cells (33.35 ± 1.8%) and H-4-11-E liver cells (46.7 ± 1.2%) at a concentration of 250 µg/mL. The binding energies of the isolated compounds for glycogen phosphorylase (1NOI) and α-amylase (1OSE) were stronger (<-8.1) than those of the positive controls. Overall, all tested compounds exhibited characteristics indicative of their potential as antidiabetic agents; however, toxicity profiling predicted epiafzelechin and epicatechin as better alternatives. The ethyl acetate fraction and its compounds, particularly epiafzelechin, showed promise as antidiabetic agents. However, further comprehensive studies are necessary to validate these findings.
Keywords: ADMET; Ficus lutea; antidiabetic; molecular docking; phytochemical.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Antidiabetic activity of the ethyl acetate fraction of Ficus lutea (Moraceae) leaf extract: comparison of an in vitro assay with an in vivo obese mouse model.BMC Complement Altern Med. 2016 Mar 31;16:110. doi: 10.1186/s12906-016-1087-z. BMC Complement Altern Med. 2016. PMID: 27029351 Free PMC article.
-
Antidiabetic potential of Catechu via assays for α-glucosidase, α-amylase, and glucose uptake in adipocytes.J Ethnopharmacol. 2022 Jun 12;291:115118. doi: 10.1016/j.jep.2022.115118. Epub 2022 Feb 21. J Ethnopharmacol. 2022. PMID: 35202712
-
Crude extract and isolated bioactive compounds from Notholirion thomsonianum (Royale) Stapf as multitargets antidiabetic agents: in-vitro and molecular docking approaches.BMC Complement Med Ther. 2021 Oct 27;21(1):270. doi: 10.1186/s12906-021-03443-7. BMC Complement Med Ther. 2021. PMID: 34706708 Free PMC article.
-
Evaluation of the inhibition of carbohydrate hydrolysing enzymes, antioxidant activity and polyphenolic content of extracts of ten African Ficus species (Moraceae) used traditionally to treat diabetes.BMC Complement Altern Med. 2013 May 4;13:94. doi: 10.1186/1472-6882-13-94. BMC Complement Altern Med. 2013. PMID: 23641947 Free PMC article.
-
A role of Ficus species in the management of diabetes mellitus: A review.J Ethnopharmacol. 2018 Apr 6;215:210-232. doi: 10.1016/j.jep.2017.12.045. Epub 2018 Jan 3. J Ethnopharmacol. 2018. PMID: 29305899 Review.
Cited by
-
In silico screening of Hippophae rhamnoides polyphenols targeting the RhoA protein as a potential liver cancer treatment.J Taibah Univ Med Sci. 2025 Feb 13;20(1):89-106. doi: 10.1016/j.jtumed.2025.01.006. eCollection 2025 Feb. J Taibah Univ Med Sci. 2025. PMID: 40034742 Free PMC article.
-
In silico drug repurposing of potential antiviral inhibitors targeting methyltransferase (2'-O-MTase) domain of Marburg virus.In Silico Pharmacol. 2025 Apr 24;13(2):70. doi: 10.1007/s40203-025-00355-z. eCollection 2025. In Silico Pharmacol. 2025. PMID: 40291443
-
Antidiabetic Activity, Phytochemical Analysis, and Acute Oral Toxicity Test of Combined Ethanolic Extract of Syzygium polyanthum and Muntingia calabura Leaves.ScientificWorldJournal. 2024 Jul 17;2024:3607396. doi: 10.1155/2024/3607396. eCollection 2024. ScientificWorldJournal. 2024. PMID: 39050386 Free PMC article.
-
In Vitro and Molecular Docking Evaluation of the Anticholinesterase and Antidiabetic Effects of Compounds from Terminalia macroptera Guill. & Perr. (Combretaceae).Molecules. 2024 May 23;29(11):2456. doi: 10.3390/molecules29112456. Molecules. 2024. PMID: 38893333 Free PMC article.
-
Assessment of the conceivable inhibitory activity of pathogenic microorganisms extracted from seaweed using phytochemicals, antioxidants, and in-silico molecular dynamic simulation.Sci Rep. 2024 Oct 5;14(1):23200. doi: 10.1038/s41598-024-70620-2. Sci Rep. 2024. PMID: 39368983 Free PMC article.
References
-
- World Health Organization . Global Status Report on Noncommunicable Diseases 2010. World Health Organization; Geneva, Switzerland: 2011.
-
- World Health Organization . Part 1: Diagnosis and Classification of Diabetes Mellitus. World Health Organization; Geneva, Switzerland: 1999. Consultation, Definition, diagnosis and classification of diabetes mellitus and its complications. Report of a WHO Consultation.
-
- Statistics South Africa . Mortality and Causes of Death in South Africa, Findings from Death Notification. Statistics South Africa; Pretoria, South Africa: 2015.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

