Integrin-linked kinase expression in myeloid cells promotes colon tumorigenesis
- PMID: 38077324
- PMCID: PMC10710162
- DOI: 10.3389/fimmu.2023.1270194
Integrin-linked kinase expression in myeloid cells promotes colon tumorigenesis
Abstract
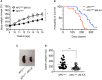
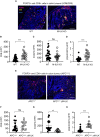
Colorectal cancer (CRC) is one of the most common forms of cancer worldwide and treatment options for advanced CRC, which has a low 5-year survival rate, remain limited. Integrin-linked kinase (ILK), a multifunctional, scaffolding, pseudo-kinase regulating many integrin-mediated cellular processes, is highly expressed in many cancers. However, the role of ILK in cancer progression is yet to be fully understood. We have previously uncovered a pro-inflammatory role for myeloid-specific ILK in dextran sodium sulfate (DSS)-induced colitis. To establish a correlation between chronic intestinal inflammation and colorectal cancer (CRC), we investigated the role of myeloid-ILK in mouse models of CRC. When myeloid-ILK deficient mice along with the WT control mice were subjected to colitis-associated and APCmin/+-driven CRC, tumour burden was reduced by myeloid-ILK deficiency in both models. The tumour-promoting phenotype of macrophages, M2 polarization, in vitro was impaired by the ILK deficiency and the number of M2-specific marker CD206-expressing tumour-associated macrophages (TAMs) in vivo were significantly diminished in myeloid-ILK deficient mice. Myeloid-ILK deficient mice showed enhanced tumour infiltration of CD8+ T cells and reduced tumour infiltration of FOXP3+ T cells in colitis-associated and APCmin/+-driven CRC, respectively, with an overall elevated CD8+/FOXP3+ ratio suggesting an anti-tumour immune phenotypes. In patient CRC tissue microarrays we observed elevated ILK+ myeloid (ILK+ CD11b+) cells in tumour sections compared to adjacent normal tissues, suggesting a conserved role for myeloid-ILK in CRC development in both human and animal models. This study identifies myeloid-specific ILK expression as novel driver of CRC, which could be targeted as a potential therapeutic option for advanced disease.
Keywords: Apc min/+; CAC; CD8; CRC; Foxp3; ILK; M2 polarization; myeloid.
Copyright © 2023 Ahmed, Almasabi, Firestein and Williams.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures





Similar articles
-
TRIB3 Interacts With β-Catenin and TCF4 to Increase Stem Cell Features of Colorectal Cancer Stem Cells and Tumorigenesis.Gastroenterology. 2019 Feb;156(3):708-721.e15. doi: 10.1053/j.gastro.2018.10.031. Epub 2018 Oct 24. Gastroenterology. 2019. PMID: 30365932
-
Integrin-Linked Kinase Expression in Myeloid Cells Promotes Inflammatory Signaling during Experimental Colitis.J Immunol. 2017 Sep 15;199(6):2128-2139. doi: 10.4049/jimmunol.1700125. Epub 2017 Aug 9. J Immunol. 2017. PMID: 28794235
-
The active fraction of Garcinia yunnanensis suppresses the progression of colorectal carcinoma by interfering with tumorassociated macrophage-associated M2 macrophage polarization in vivo and in vitro.FASEB J. 2020 Jun;34(6):7387-7403. doi: 10.1096/fj.201903011R. Epub 2020 Apr 13. FASEB J. 2020. PMID: 32283574
-
Integrin-linked kinase regulates cell proliferation and tumour growth in murine colitis-associated carcinogenesis.Gut. 2008 Jul;57(7):931-40. doi: 10.1136/gut.2007.142778. Epub 2008 Mar 7. Gut. 2008. PMID: 18326560
-
A Potential Role for Integrin-Linked Kinase in Colorectal Cancer Growth and Progression via Regulating Senescence and Immunity.Front Genet. 2021 Jun 7;12:638558. doi: 10.3389/fgene.2021.638558. eCollection 2021. Front Genet. 2021. PMID: 34163519 Free PMC article. Review.
Cited by
-
Exosomal miR-106a-5p from highly metastatic colorectal cancer cells drives liver metastasis by inducing macrophage M2 polarization in the tumor microenvironment.J Exp Clin Cancer Res. 2024 Oct 9;43(1):281. doi: 10.1186/s13046-024-03204-7. J Exp Clin Cancer Res. 2024. PMID: 39385295 Free PMC article.
-
The integrin adhesome and control of anti-tumour immunity.Biochem Soc Trans. 2024 Dec 19;52(6):2455-2468. doi: 10.1042/BST20240386. Biochem Soc Trans. 2024. PMID: 39641590 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

