Prevention of respiratory virus transmission by resident memory CD8+ T cells
- PMID: 38086420
- PMCID: PMC11040656
- DOI: 10.1038/s41586-023-06937-1
Prevention of respiratory virus transmission by resident memory CD8+ T cells
Abstract
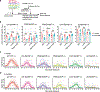
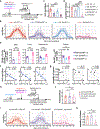
An ideal vaccine both attenuates virus growth and disease in infected individuals and reduces the spread of infections in the population, thereby generating herd immunity. Although this strategy has proved successful by generating humoral immunity to measles, yellow fever and polio, many respiratory viruses evolve to evade pre-existing antibodies1. One approach for improving the breadth of antiviral immunity against escape variants is through the generation of memory T cells in the respiratory tract, which are positioned to respond rapidly to respiratory virus infections2-6. However, it is unknown whether memory T cells alone can effectively surveil the respiratory tract to the extent that they eliminate or greatly reduce viral transmission following exposure of an individual to infection. Here we use a mouse model of natural parainfluenza virus transmission to quantify the extent to which memory CD8+ T cells resident in the respiratory tract can provide herd immunity by reducing both the susceptibility of acquiring infection and the extent of transmission, even in the absence of virus-specific antibodies. We demonstrate that protection by resident memory CD8+ T cells requires the antiviral cytokine interferon-γ (IFNγ) and leads to altered transcriptional programming of epithelial cells within the respiratory tract. These results suggest that tissue-resident CD8+ T cells in the respiratory tract can have important roles in protecting the host against viral disease and limiting viral spread throughout the population.
© 2023. The Author(s), under exclusive licence to Springer Nature Limited.
Conflict of interest statement
Competing interests
Authors declare that they have no competing interests.
Figures







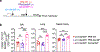




References
Methods references
-
- Upadhyay R, Boiarsky JA, Pantsulaia G, Svensson-Arvelund J, Lin MJ, Wroblewska A, Bhalla S, Scholler N, Bot A, Rossi JM, Sadek N, Parekh S, Lagana A, Baccarini A, Merad M, Brown BD, Brody JD. A Critical Role for Fas-Mediated Off-Target Tumor Killing in T-cell Immunotherapy. Cancer Discov. 2021. Mar;11(3):599–613. doi: 10.1158/2159-8290.CD-20-0756. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

