Rapid characterization of adeno-associated virus (AAV) capsid proteins using microchip ZipChip CE-MS
- PMID: 38102410
- PMCID: PMC10800304
- DOI: 10.1007/s00216-023-05097-5
Rapid characterization of adeno-associated virus (AAV) capsid proteins using microchip ZipChip CE-MS
Abstract
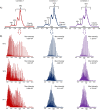
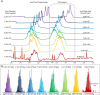
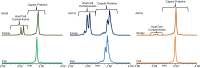
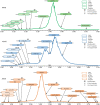
Adeno-associated viruses (AAVs) are viral vectors used as delivery systems for gene therapies. Intact protein characterization of AAV viral capsid proteins (VPs) and their post-translational modifications is critical to ensuring product quality. In this study, microchip-based ZipChip capillary electrophoresis-mass spectrometry (CE-MS) was applied for the rapid characterization of AAV intact VPs, specifically full and empty viral capsids of serotypes AAV6, AAV8 and AAV9, which was accomplished using 5 min of analysis time. Low levels of dimethyl sulfoxide (4%) in the background electrolyte (BGE) improved MS signal quality and component detection. A sensitivity evaluation revealed consistent detection of VP proteoforms when as little as 2.64 × 106 viral particles (≈26.4 picograms) were injected. Besides the traditional VP proteoforms used for serotype identification, multiple VP3 variants were detected, including truncated VP3 variants most likely generated by leaky scanning as well as unacetylated and un-cleaved VP3 proteoforms. Phosphorylation, known to impact AAV transduction efficiency, was also seen in all serotypes analysed. Additionally, low abundant fragments originating from either N- or C-terminus truncation were detected. As the aforementioned VP components can impact product quality and efficacy, the ZipChip's ability to rapidly characterize them illustrates its strength in monitoring product quality during AAV production.
Keywords: Adeno-associated virus; Characterization; Gene therapy; Microchip CE-MS; Post translation modifications; Product quality monitoring.
© 2023. The Author(s).
Conflict of interest statement
AK, ER, and KY are employed by 908 Devices, the company that develops and produces the ZipChip hardware and consumables that was used during the study. JB received funding and instrument access from 908 Devices that was used to support the research activity reported herein. JS and SC used the materials provided by 908 Devices in conducting the research but have no financial conflict of interest to report. Other than those disclosed here, the authors declare that the research was conducted in the absence of any commercial, financial, or nonfinancial relationships that could be construed as a potential conflict of interest.
Figures
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

