On-off conduction photoswitching in modelled spiropyran-based metal-organic frameworks
- PMID: 38110545
- PMCID: PMC10728195
- DOI: 10.1038/s42004-023-01072-4
On-off conduction photoswitching in modelled spiropyran-based metal-organic frameworks
Abstract
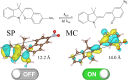
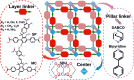
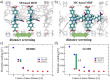
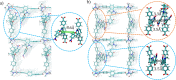
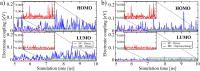
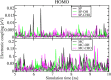
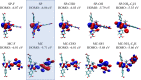
Materials with photoswitchable electronic properties and conductance values that can be reversibly changed over many orders of magnitude are highly desirable. Metal-organic framework (MOF) films functionalized with photoresponsive spiropyran molecules demonstrated the general possibility to switch the conduction by light with potentially large on-off-ratios. However, the fabrication of MOF materials in a trial-and-error approach is cumbersome and would benefit significantly from in silico molecular design. Based on the previous proof-of-principle investigation, here, we design photoswitchable MOFs which incorporate spiropyran photoswitches at controlled positions with defined intermolecular distances and orientations. Using multiscale modelling and automated workflow protocols, four MOF candidates are characterized and their potential for photoswitching the conductivity is explored. Using ab initio calculations of the electronic coupling between the molecules in the MOF, we show that lattice distances and vibrational flexibility tremendously modulate the possible conduction photoswitching between spiropyran- and merocyanine-based MOFs upon light absorption, resulting in average on-off ratios higher than 530 and 4200 for p- and n-conduction switching, respectively. Further functionalization of the photoswitches with electron-donating/-withdrawing groups is demonstrated to shift the energy levels of the frontier orbitals, permitting a guided design of new spiropyran-based photoswitches towards controlled modification between electron and hole conduction in a MOF.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interest.
Figures
Similar articles
-
Conductance Photoswitching of Metal-Organic Frameworks with Embedded Spiropyran.Angew Chem Int Ed Engl. 2019 Jan 21;58(4):1193-1197. doi: 10.1002/anie.201811458. Epub 2018 Dec 20. Angew Chem Int Ed Engl. 2019. PMID: 30421842
-
Solution-Like Behavior of Photoswitchable Spiropyrans Embedded in Metal-Organic Frameworks.Inorg Chem. 2017 Nov 6;56(21):13100-13110. doi: 10.1021/acs.inorgchem.7b01908. Epub 2017 Oct 11. Inorg Chem. 2017. PMID: 29019664
-
A Light-Responsive Metal-Organic Framework Hybrid Membrane with High On/Off Photoswitchable Proton Conductivity.Angew Chem Int Ed Engl. 2020 May 11;59(20):7732-7737. doi: 10.1002/anie.202002389. Epub 2020 Mar 13. Angew Chem Int Ed Engl. 2020. PMID: 32090427
-
Advanced Photoresponsive Materials Using the Metal-Organic Framework Approach.Adv Mater. 2020 May;32(20):e1905227. doi: 10.1002/adma.201905227. Epub 2019 Nov 25. Adv Mater. 2020. PMID: 31763731 Review.
-
Stimuli-Responsive Metal-Organic Frameworks with Photoswitchable Azobenzene Side Groups.Macromol Rapid Commun. 2018 Jan;39(1). doi: 10.1002/marc.201700239. Epub 2017 Jul 31. Macromol Rapid Commun. 2018. PMID: 28758288 Review.
Cited by
-
Modified Working Electrodes for Organic Electrosynthesis.ACS Org Inorg Au. 2024 Oct 2;4(6):579-603. doi: 10.1021/acsorginorgau.4c00050. eCollection 2024 Dec 4. ACS Org Inorg Au. 2024. PMID: 39649987 Free PMC article. Review.
-
Engineering Photoswitching Dynamics in 3D Photochromic Metal-Organic Frameworks through a Metal-Organic Polyhedron Design.J Am Chem Soc. 2025 Mar 12;147(10):8568-8577. doi: 10.1021/jacs.4c17203. Epub 2025 Feb 25. J Am Chem Soc. 2025. PMID: 39995312 Free PMC article.
-
Supramolecular systems and their connection with metal-organic structures.Front Chem. 2024 Nov 5;12:1468916. doi: 10.3389/fchem.2024.1468916. eCollection 2024. Front Chem. 2024. PMID: 39564433 Free PMC article. Review.
-
Illuminating Biomimetic Nanochannels: Unveiling Macroscopic Anticounterfeiting and Photoswitchable Ion Conductivity via Polymer Tailoring.ACS Nano. 2024 Oct 1;18(39):26948-26960. doi: 10.1021/acsnano.4c08801. Epub 2024 Sep 20. ACS Nano. 2024. PMID: 39302690 Free PMC article.
-
The Effect of Aldehyde and Carboxylic Acid Substitution on the Isomerization of Hemithioindigo Photoswitches.Chemistry. 2025 Jun 3;31(31):e202501108. doi: 10.1002/chem.202501108. Epub 2025 May 2. Chemistry. 2025. PMID: 40243361 Free PMC article.
References
-
- Browne, W. R. & Feringa, B. L. Chiroptical molecular switches. Molecular Switches 121–179 (John Wiley & Sons, Ltd), 10.1002/9783527634408.ch5 (2011).
-
- Paramonov SV, Lokshin V, Fedorova OA. Spiropyran, chromene or spirooxazine ligands: Insights into mutual relations between complexing and photochromic properties. J. Photochem. Photobiol. C Photochem. Rev. 2011;12:209–236. doi: 10.1016/j.jphotochemrev.2011.09.001. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

