Nano-Regulator Inhibits Tumor Immune Escape via the "Two-Way Regulation" Epigenetic Therapy Strategy
- PMID: 38110834
- PMCID: PMC10916662
- DOI: 10.1002/advs.202305275
Nano-Regulator Inhibits Tumor Immune Escape via the "Two-Way Regulation" Epigenetic Therapy Strategy
Abstract
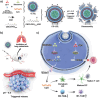
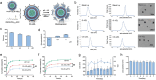
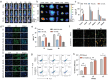
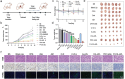
Tumor immune escape caused by low levels of tumor immunogenicity and immune checkpoint-dependent suppression limits the immunotherapeutic effect. Herein, a "two-way regulation" epigenetic therapeutic strategy is proposed using a novel nano-regulator that inhibits tumor immune escape by upregulating expression of tumor-associated antigens (TAAs) to improve immunogenicity and downregulating programmed cell death 1 ligand 1 (PD-L1) expression to block programmed death-1 (PD-1)/PD-L1. To engineer the nano-regulator, the DNA methyltransferase (DNMT) inhibitor zebularine (Zeb) and the bromodomain-containing protein 4 (BRD4) inhibitor JQ1 are co-loaded into the cationic liposomes with condensing the toll-like receptor 9 (TLR9) agonist cytosine-phosphate-guanine (CpG) via electrostatic interactions to obtain G-J/ZL. Then, asparagine-glycine-arginine (NGR) modified material carboxymethyl-chitosan (CMCS) is coated on the surface of G-J/ZL to construct CG-J/ZL. CG-J/ZL is shown to target tumor tissue and disassemble under the acidic tumor microenvironment (TME). Zeb upregulated TAAs expression to improve the immunogenicity; JQ1 inhibited PD-L1 expression to block immune checkpoint; CpG promote dendritic cell (DC) maturation and reactivated the ability of tumour-associated macrophages (TAM) to kill tumor cells. Taken together, these results demonstrate that the nano-regulator CG-J/ZL can upregulate TAAs expression to enhance T-cell infiltration and downregulate PD-L1 expression to improve the recognition of tumor cells by T-cells, representing a promising strategy to improve antitumor immune response.
Keywords: epigenetic regulation; nano-regulator; tumor immune escape; two-way regulation.
© 2023 The Authors. Advanced Science published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
BRD4 promotes immune escape of glioma cells by upregulating PD-L1 expression.J Neurooncol. 2025 Feb;171(3):669-679. doi: 10.1007/s11060-024-04889-8. Epub 2024 Nov 28. J Neurooncol. 2025. PMID: 39607572
-
WDR5-H3K4me3 epigenetic axis regulates OPN expression to compensate PD-L1 function to promote pancreatic cancer immune escape.J Immunother Cancer. 2021 Jul;9(7):e002624. doi: 10.1136/jitc-2021-002624. J Immunother Cancer. 2021. PMID: 34326167 Free PMC article.
-
Combination of epigenetic regulation with gene therapy-mediated immune checkpoint blockade induces anti-tumour effects and immune response in vivo.Nat Commun. 2021 Nov 18;12(1):6742. doi: 10.1038/s41467-021-27078-x. Nat Commun. 2021. PMID: 34795289 Free PMC article.
-
Epigenetic abnormalities of classical Hodgkin lymphoma and its effect on immune escape.Cell Biochem Funct. 2020 Apr;38(3):242-248. doi: 10.1002/cbf.3463. Epub 2019 Nov 11. Cell Biochem Funct. 2020. PMID: 31709594 Review.
-
Role of the tumor microenvironment in PD-L1/PD-1-mediated tumor immune escape.Mol Cancer. 2019 Jan 15;18(1):10. doi: 10.1186/s12943-018-0928-4. Mol Cancer. 2019. PMID: 30646912 Free PMC article. Review.
Cited by
-
Intrinsic PD-L1 Degradation Induced by a Novel Self-Assembling Hexapeptide for Enhanced Cancer Immunotherapy.Adv Sci (Weinh). 2025 Jan;12(2):e2410145. doi: 10.1002/advs.202410145. Epub 2024 Nov 12. Adv Sci (Weinh). 2025. PMID: 39530653 Free PMC article.
-
Reprogramming Dysfunctional Dendritic Cells by a Versatile Catalytic Dual Oxide Antigen-Captured Nanosponge for Remotely Enhancing Lung Metastasis Immunotherapy.ACS Nano. 2025 Jan 21;19(2):2117-2135. doi: 10.1021/acsnano.4c09525. Epub 2024 Dec 31. ACS Nano. 2025. PMID: 39739571 Free PMC article.
-
DNMT1 promotes bladder cancer progression and immune escape by inhibiting MYH11 expression by methylating its promoter.Int Urol Nephrol. 2025 May 2. doi: 10.1007/s11255-025-04527-w. Online ahead of print. Int Urol Nephrol. 2025. PMID: 40314887
-
Engineered Exosomes Biopotentiated Hydrogel Promote Hair Follicle Growth via Reprogramming the Perifollicular Microenvironment.Pharmaceutics. 2024 Jul 13;16(7):935. doi: 10.3390/pharmaceutics16070935. Pharmaceutics. 2024. PMID: 39065633 Free PMC article.
References
-
- Bagchi S., Yuan R., Engleman E. G., Annu. Rev. Pathol.: Mech. Dis. 2021, 16, 223. - PubMed
-
- Thorsson V., Gibbs D. L., Brown S. D., Wolf D., Bortone D. S., Ou Yang T.‐H., Porta‐Pardo E., Gao G. F., Plaisier C. L., Eddy J. A., Ziv E., Culhane A. C., Paull E. O., Sivakumar I. K. A, Gentles A. J., Malhotra R., Farshidfar F., Colaprico A., Parker J. S., Mose L. E., Vo N. S., Liu J., Liu Y., Rader J., Dhankani V., Reynolds S. M., Bowlby R., Califano A., Cherniack A. D., Anastassiou D., et al., Immunity 2018, 48, 812. - PMC - PubMed
-
- Hao Y., Li H., Ge X., Liu Y., Li X., Liu Y., Chen H., Zhang S., Zou J., Huang L., Zhao F., Kang D., De Geest B. G., Zhang Z., Angew. Chem. 2022, 61, e202214992. - PubMed
-
- Lu M., Xing H. N., Shao W. X., Zhang T., Zhang M. J., Wang Y. C., Li F. Z., Weng Y. H., Zheng A. P., Huang Y. Y., Liang X. J., Adv. Mater. 2022, 34. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
