GPRC5C regulates the composition of cilia in the olfactory system
- PMID: 38110903
- PMCID: PMC10729543
- DOI: 10.1186/s12915-023-01790-0
GPRC5C regulates the composition of cilia in the olfactory system
Abstract
Background: Olfactory sensory neurons detect odourants via multiple long cilia that protrude from their dendritic endings. The G protein-coupled receptor GPRC5C was identified as part of the olfactory ciliary membrane proteome, but its function and localization is unknown.
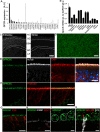
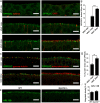
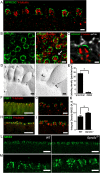
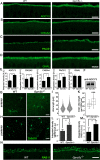
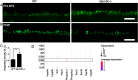
Results: High-resolution confocal and electron microscopy revealed that GPRC5C is located at the base of sensory cilia in olfactory neurons, but not in primary cilia of immature neurons or stem cells. Additionally, GPRC5C localization in sensory cilia parallels cilia formation and follows the formation of the basal body. In closer examination, GPRC5C was found in the ciliary transition zone. GPRC5C deficiency altered the structure of sensory cilia and increased ciliary layer thickness. However, primary cilia were unaffected. Olfactory sensory neurons from Gprc5c-deficient mice exhibited altered localization of olfactory signalling cascade proteins, and of ciliary phosphatidylinositol-4,5-bisphosphat. Sensory neurons also exhibited increased neuronal activity as well as altered mitochondrial morphology, and knockout mice had an improved ability to detect food pellets based on smell.
Conclusions: Our study shows that GPRC5C regulates olfactory cilia composition and length, thereby controlling odour perception.
Keywords: Cilia; GPRC5C; Olfactory; Sensory; Smell.
© 2023. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

