Multimodal analysis in symptomatic MIDD-associated retinopathy. A case report and literature review
- PMID: 38111473
- PMCID: PMC10726563
- DOI: 10.3205/oc000231
Multimodal analysis in symptomatic MIDD-associated retinopathy. A case report and literature review
Abstract
Purpose: To present results of contemporary multimodal ophthalmic imaging in a case of maternally inherited diabetes and deafness (MIDD) and a literature review of MIDD.
Methods: A case of a 47-year-old female with diabetes mellitus, severe insulin resistance, familial lipodystrohy, deafness and increasing problems with vision is reported. A full ophthalmic examination was done, including best corrected visual acuity (BCVA, LogMAR), funduscopy, and imaging studies: optical coherence tomography (OCT), OCT angiography (OCT-A), fundus autofloresence (FAF), visual fields (HVF) 10-2 , electrophysiology (EP) and genetic testing were performed. Literature available on the topic was reviewed.
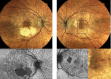
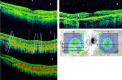
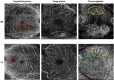
Results: BCVA was 0.06 LogMAR in the right eye and 0.1 LogMAR in the left. Funduscopy revealed atrophy (AT) and pigmentary changes but no diabetic retinopathy. HVF confirmed corresponding defects. The imaging and diagnostic tests showed the following abnormalities: FAF: hypoautofluoresence in areas of AT and mottled appearance in the macular and peripapillary area; OCT: attenuation of outer retinal layers and retinal pigment epithelium (RPE) in the AT; OCT-A: thinning of the deep capillary plexus and choriocapillaris; EP: abnormalities on full field electroretinogram (ERG), 30 Hz flicker and single cone flash response; multifocal ERG: reduced responses; genetic testing: A-to-G transition mutation at position 3243 of the mitochondrial genome, typical for MIDD. After one year OCT ganglion cell analysis showed loss of thickness.
Conclusions: Genetic testing should be considered in diabetic patients with pigmentary retinopathy. Imaging studies and diagnostic testing showed structural and functional retinal changes, confined to the macula and progressive in nature.
Keywords: OCT; OCT-angiography; diabetes mellitus; fundus autofluorescence; maternally inherited diabetes and deafness; mitochondrial disease; multimodal imaging; noninsulin-dependent diabetes mellitus with deafness; pigmentary retinopathy; retinal ganglion cell layer.
Copyright © 2023 Chwiejczak et al.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures



References
-
- OMIM® – Online Mendelian Inheritance in Man®. # 520000: Diabetes and deafness, maternally inherited; MIDD. Available from: https://www.omim.org/entry/520000.
-
- van den Ouweland JM, Lemkes HH, Ruitenbeek W, Sandkuijl LA, de Vijlder MF, Struyvenberg PA, van de Kamp JJ, Maassen JA. Mutation in mitochondrial tRNA(Leu)(UUR) gene in a large pedigree with maternally transmitted type II diabetes mellitus and deafness. Nat Genet. 1992 Aug;1(5):368–371. doi: 10.1038/ng0892-368. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources

