ATP-mediated increase in H+ efflux from retinal Müller cells of the axolotl
- PMID: 38116604
- PMCID: PMC11286307
- DOI: 10.1152/jn.00321.2023
ATP-mediated increase in H+ efflux from retinal Müller cells of the axolotl
Abstract
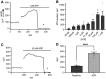
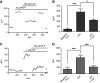
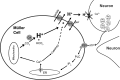
Previous work has shown that activation of tiger salamander retinal radial glial cells by extracellular ATP induces a pronounced extracellular acidification, which has been proposed to be a potent modulator of neurotransmitter release. This study demonstrates that low micromolar concentrations of extracellular ATP similarly induce significant H+ effluxes from Müller cells isolated from the axolotl retina. Müller cells were enzymatically isolated from axolotl retina and H+ fluxes were measured from individual cells using self-referencing H+-selective microelectrodes. The increased H+ efflux from axolotl Müller cells induced by extracellular ATP required activation of metabotropic purinergic receptors and was dependent upon calcium released from internal stores. We further found that the ATP-evoked increase in H+ efflux from Müller cells of both tiger salamander and axolotl were sensitive to pharmacological agents known to interrupt calmodulin and protein kinase C (PKC) activity: chlorpromazine (CLP), trifluoperazine (TFP), and W-7 (all calmodulin inhibitors) and chelerythrine, a PKC inhibitor, all attenuated ATP-elicited increases in H+ efflux. ATP-initiated H+ fluxes of axolotl Müller cells were also significantly reduced by amiloride, suggesting a significant contribution by sodium-hydrogen exchangers (NHEs). In addition, α-cyano-4-hydroxycinnamate (4-cin), a monocarboxylate transport (MCT) inhibitor, also reduced the ATP-induced increase in H+ efflux in both axolotl and tiger salamander Müller cells, and when combined with amiloride, abolished ATP-evoked increase in H+ efflux. These data suggest that axolotl Müller cells are likely to be an excellent model system to understand the cell-signaling pathways regulating H+ release from glia and the role this may play in modulating neuronal signaling.NEW & NOTEWORTHY Glial cells are a key structural part of the tripartite synapse and have been suggested to regulate synaptic transmission, but the regulatory mechanisms remain unclear. We show that extracellular ATP, a potent glial cell activator, induces H+ efflux from axolotl retinal Müller (glial) cells through a calcium-dependent pathway that is likely to involve calmodulin, PKC, Na+/H+ exchange, and monocarboxylate transport, and suggest that such H+ release may play a key role in modulating neuronal transmission.
Keywords: calmodulin; glia; monocarboxylate transport; pH; protein kinase C.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures







References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

