NLRP3 inflammasome activation and symptom burden in KRAS-mutated CMML patients is reverted by IL-1 blocking therapy
- PMID: 38118408
- PMCID: PMC10772462
- DOI: 10.1016/j.xcrm.2023.101329
NLRP3 inflammasome activation and symptom burden in KRAS-mutated CMML patients is reverted by IL-1 blocking therapy
Abstract
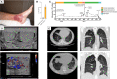
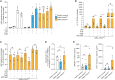
Chronic myelomonocytic leukemia (CMML) is frequently associated with mutations in the rat sarcoma gene (RAS), leading to worse prognosis. RAS mutations result in active RAS-GTP proteins, favoring myeloid cell proliferation and survival and inducing the NLRP3 inflammasome together with the apoptosis-associated speck-like protein containing a caspase recruitment domain (ASC), which promote caspase-1 activation and interleukin (IL)-1β release. Here, we report, in a cohort of CMML patients with mutations in KRAS, a constitutive activation of the NLRP3 inflammasome in monocytes, evidenced by ASC oligomerization and IL-1β release, as well as a specific inflammatory cytokine signature. Treatment of a CMML patient with a KRASG12D mutation using the IL-1 receptor blocker anakinra inhibits NLRP3 inflammasome activation, reduces monocyte count, and improves the patient's clinical status, enabling a stem cell transplant. This reveals a basal inflammasome activation in RAS-mutated CMML patients and suggests potential therapeutic applications of NLRP3 and IL-1 blockers.
Keywords: CMML; IL-1 blockers; KRAS; NLRP3 blockers; NLRP3 inflammasome; RAS mutations; anakinra; inflammation; myelodysplastic syndromes; myeloproliferative neoplasms.
Copyright © 2023 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests P.P. is consultant of Viva In Vitro Diagnostics SL. P.P., H.M.-B., and C.G.-P. are inventors on patent PCT/EP2020/056729. L.H.-N., L.M.-A., H.M.-B., C.G.-P., and P.P. are co-founders of Viva In Vitro Diagnostics SL but declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Arber D.A., Orazi A., Hasserjian R.,P., Borowitz M.J., Calvo K.R., Kvasnicka H.M., Wang S.A., Bagg A., Barbui T., Branford S., et al. International Consensus Classification of Myeloid Neoplasms and Acute Leukemias: integrating morphologic, clinical, and genomic data. Blood. 2022;140:1200–1228. - PMC - PubMed
-
- Khoury J.D., Solary E., Abla O., Akkari Y., Alaggio R., Apperley J.F., Bejar R., Berti E., Busque L., Chan J.K.C., et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia. 2022;36:1703–1719. - PMC - PubMed
-
- Kohlmann A., Grossmann V., Klein H.U., Schindela S., Weiss T., Kazak B., Dicker F., Schnittger S., Dugas M., Kern W., et al. Next-Generation Sequencing Technology Reveals a Characteristic Pattern of Molecular Mutations in 72.8% of Chronic Myelomonocytic Leukemia by Detecting Frequent Alterations in TET2 , CBL , RAS , and RUNX1. J. Clin. Oncol. 2010;28:3858–3865. - PubMed

