Acetylshikonin induces necroptosis via the RIPK1/RIPK3-dependent pathway in lung cancer
- PMID: 38126996
- PMCID: PMC10781480
- DOI: 10.18632/aging.205316
Acetylshikonin induces necroptosis via the RIPK1/RIPK3-dependent pathway in lung cancer
Abstract
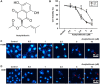
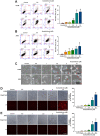
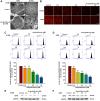
Despite advances in therapeutic strategies, lung cancer remains the leading cause of cancer-related death worldwide. Acetylshikonin is a derivative of the traditional Chinese medicine Zicao and presents a variety of anticancer properties. However, the effects of acetylshikonin on lung cancer have not been fully understood yet. This study explored the mechanisms underlying acetylshikonin-induced cell death in non-small cell lung cancer (NSCLC). Treating NSCLC cells with acetylshikonin significantly reduced cell viability, as evidenced by chromatin condensation and the appearance of cell debris. Acetylshikonin has also been shown to increase cell membrane permeability and induce cell swelling, leading to an increase in the population of necrotic cells. When investigating the mechanisms underlying acetylshikonin-induced cell death, we discovered that acetylshikonin promoted oxidative stress, decreased mitochondrial membrane potential, and promoted G2/M phase arrest in lung cancer cells. The damage to NSCLC cells induced by acetylshikonin resembled results involving alterations in the cell membrane and mitochondrial morphology. Our analysis of oxidative stress revealed that acetylshikonin induced lipid oxidation and down-regulated the expression of glutathione peroxidase 4 (GPX4), which has been associated with necroptosis. We also determined that acetylshikonin induces the phosphorylation of receptor-interacting serine/threonine-protein kinase 1 (RIPK1)/RIPK3 and mixed lineage kinase domain-like kinase (MLKL). Treatment with RIPK1 inhibitors (necrostatin-1 or 7-Cl-O-Nec-1) significantly reversed acetylshikonin-induced MLKL phosphorylation and NSCLC cell death. These results indicate that acetylshikonin activated the RIPK1/RIPK3/MLKL cascade, leading to necroptosis in NSCLC cells. Our findings indicate that acetylshikonin reduces lung cancer cells by promoting G2/M phase arrest and necroptosis.
Keywords: ROS; acetylshikonin; human lung cancer; necroptosis.
Conflict of interest statement
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

