Cortical effects of wrist tendon vibration during an arm tracking task in chronic stroke survivors: An EEG study
- PMID: 38127998
- PMCID: PMC10735026
- DOI: 10.1371/journal.pone.0266586
Cortical effects of wrist tendon vibration during an arm tracking task in chronic stroke survivors: An EEG study
Abstract
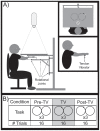
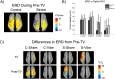
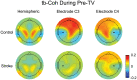
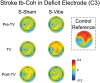
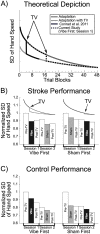
The purpose of this study was to characterize changes in cortical activity and connectivity in stroke survivors when vibration is applied to the wrist flexor tendons during a visuomotor tracking task. Data were collected from 10 chronic stroke participants and 10 neurologically-intact controls while tracking a target through a figure-8 pattern in the horizontal plane. Electroencephalography (EEG) was used to measure cortical activity (beta band desynchronization) and connectivity (beta band task-based coherence) with movement kinematics and performance error also being recorded during the task. All participants came into our lab on two separate days and performed three blocks (16 trials each, 48 total trials) of tracking, with the middle block including vibration or sham applied at the wrist flexor tendons. The order of the sessions (Vibe vs. Sham) was counterbalanced across participants to prevent ordering effects. During the Sham session, cortical activity increased as the tracking task progressed (over blocks). This effect was reduced when vibration was applied to controls. In contrast, vibration increased cortical activity during the vibration period in participants with stroke. Cortical connectivity increased during vibration, with larger effect sizes in participants with stroke. Changes in tracking performance, standard deviation of hand speed, were observed in both control and stroke groups. Overall, EEG measures of brain activity and connectivity provided insight into effects of vibration on brain control of a visuomotor task. The increases in cortical activity and connectivity with vibration improved patterns of activity in people with stroke. These findings suggest that reactivation of normal cortical networks via tendon vibration may be useful during physical rehabilitation of stroke patients.
Copyright: © 2023 Snyder et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



Similar articles
-
Effects of wrist tendon vibration on arm tracking in people poststroke.J Neurophysiol. 2011 Sep;106(3):1480-8. doi: 10.1152/jn.00404.2010. Epub 2011 Jun 22. J Neurophysiol. 2011. PMID: 21697444 Free PMC article.
-
Electroencephalography resting-state networks in people with Stroke.Brain Behav. 2021 May;11(5):e02097. doi: 10.1002/brb3.2097. Epub 2021 Mar 23. Brain Behav. 2021. PMID: 33759382 Free PMC article.
-
Effect of Tendon Vibration on Hemiparetic Arm Stability in Unstable Workspaces.PLoS One. 2015 Dec 3;10(12):e0144377. doi: 10.1371/journal.pone.0144377. eCollection 2015. PLoS One. 2015. PMID: 26633892 Free PMC article.
-
Upper limb tendon/muscle vibration in persons with subacute and chronic stroke: a systematic review and meta-analysis.Eur J Phys Rehabil Med. 2019 Oct;55(5):558-569. doi: 10.23736/S1973-9087.19.05605-3. Epub 2019 Mar 11. Eur J Phys Rehabil Med. 2019. PMID: 30868835
-
Cortical connectivity in stroke using signals from resting-state EEG: a review of current literature.Acta Neurol Belg. 2023 Apr;123(2):351-357. doi: 10.1007/s13760-022-02102-z. Epub 2022 Oct 3. Acta Neurol Belg. 2023. PMID: 36190646 Review.
Cited by
-
Distinct and additive effects of visual and vibratory feedback for motor rehabilitation: an EEG study in healthy subjects.J Neuroeng Rehabil. 2024 Sep 12;21(1):158. doi: 10.1186/s12984-024-01453-3. J Neuroeng Rehabil. 2024. PMID: 39267092 Free PMC article.
References
-
- Centers for Disease Control and Prevention (CDC). Prevalence and most common causes of disability among adults—United States, 2005. MMWR Morb Mortal Wkly Rep. 2009. May 1;58(16):421–6. - PubMed
-
- Brunnstrom S. Movement therapy in hemiplegia: a neurophysiological approach. New York: Harper & Row; 1970.
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

