Calcium Activation of the Androgen Receptor in Prostate Cells
- PMID: 38131032
- PMCID: PMC10733593
- DOI: 10.1155/2023/9907948
Calcium Activation of the Androgen Receptor in Prostate Cells
Abstract
Background: Although prostate cancer patients initially respond to androgen deprivation therapy, most patients progress to a resistant phenotype. Castration resistance is due, in part, to intratumoral and/or adrenal synthesis of androgens, overexpression or mutation of the androgen receptor (AR), stabilization of AR by chaperones, and ligand-independent activation of AR. Increasing evidence also links disruption of calcium homeostasis to progression of prostate cancer. Our previous study shows that heavy metal cadmium activates the AR through a ligand-independent mechanism. Cadmium mimics calcium in biological systems due to their similar ionic charge and radius. This study determines whether calcium activates AR and whether first- and second-generation antiandrogens block the ability of calcium to activate the receptor.
Methods: The expression of androgen-responsive genes and calcium channels was measured in prostate cells using a quantitative real-time polymerase chain reaction assay. Cell growth was measured.
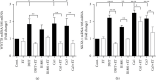
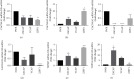
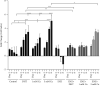
Results: To ask whether calcium activates AR, prostate cells were treated with calcium in the absence and presence of the first-generation antiandrogens hydroxyflutamide and bicalutamide and the second-generation antiandrogen enzalutamide, and the expression of androgen-responsive genes and cell growth was measured. In the normal PWR-1E cells and HEK293T cells transiently expressing AR, treatment with calcium increased the expression of androgen-responsive genes by approximately 3-fold. The increase was blocked by enzalutamide but was not consistently blocked by the first-generation antiandrogens. In LNCaP cells which contain a mutant AR, treatment with calcium also increased the expression of androgen-responsive genes by approximately 3-fold, and the increase was more effectively blocked by enzalutamide than by hydroxyflutamide or bicalutamide. Treatment with calcium also increased cell growth that was blocked by enzalutamide. To ask whether dysregulation of calcium channels is associated with castration resistance, calcium channels were measured in the normal PWR-1E prostate cells, the hormone-responsive LNCaP cells, and the castration-resistant VCaP and 22RV1 cells. Compared to normal prostate cells, the hormone-responsive and hormone-resistant cells overexpressed several calcium channels.
Conclusions: The results of this study show that calcium activates AR and increases cell growth and that calcium channels are overexpressed in hormone-responsive and hormone-resistant prostate cancer cells. Taken together, the results suggest a novel role of calcium in the castration-resistant phenotype.
Copyright © 2023 Zeina W. Sharawi et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Figures


References
-
- Kolvenbag G. J., Furr B. J. Relative potency of bicalutamide (Casodex) and flutamide (Eulexin) Urology . 1999;54(1):194–197. - PubMed
-
- Wirth M. P., See W. A., McLeod D. G., et al. Bicalutamide 150 mg in addition to standard care in patients with localized or locally advanced prostate cancer: results from the second analysis of the early prostate cancer program at median followup of 5.4 years. The Journal of Urology . 2004;172(5):1865–1870. doi: 10.1097/01.ju.0000140159.94703.80. - DOI - PubMed
-
- Fenton M. A., Shuster T. D., Fertig A. M., et al. Functional characterization of mutant androgen receptors from androgen-independent prostate cancer. Clinical Cancer Research . 1997;3(8):1383–1388. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

