Drug-Free Noninvasive Thermal Nerve Block: Validation of Sham Devices
- PMID: 38137166
- PMCID: PMC10741966
- DOI: 10.3390/brainsci13121718
Drug-Free Noninvasive Thermal Nerve Block: Validation of Sham Devices
Abstract
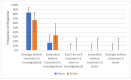
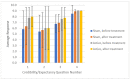
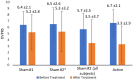
Headache is a leading cause of disability and suffering. One major challenge in developing device treatments is demonstrating their efficacy given devices' often-high placebo rate. This paper reviews the importance of validating sham devices as part of finalizing the design for larger-scale prospective randomized controlled trials in patients with chronic headache as well as the results of a prospective, single-blind trial to validate two potential sham noninvasive thermal nerve block devices. Study participants were trained to self-administer thermal nerve block treatment using sham devices in an office visit. Two different sham systems with different temperature profiles were assessed. Devices were offered for patients to use daily at-home for one week to assess the durability of sham placebo effects before participants were given active treatment in a second office visit followed by another optional week of self-administered active treatment at-home use. Sham treatments reduced pain scores by an average of 31% from 6.0 ± 2.3 to 4.3 ± 3.3, including two participants who fell asleep during the in-office treatment and woke up with no pain, but whose pain recurred after returning home during at-home use of the sham system. In-office active treatments reduced pain scores by 52% from 6.7 ± 2.1 to 3.3 ± 2.9 with sustained pain relief during optional at-home use. Successful blinding for the study was confirmed with an ideal Bang's Blinding Index of 0 and an ideal James' Blinding Index of 1. Both the sham and active treatments were viewed by participants as highly credible, and credibility increased from the beginning to end of sham treatments on average.
Keywords: chronic pain; drug-free nerve block; sham device; thermal nerve block.
Conflict of interest statement
Dr. Fishman is an equity holder/advisor in Thermaquil relevant to the paper. Dr. Chitneni has no conflict of interest to disclose for this work. Dr. Abd-Elsayed is an equity holder/advisor in Thermaquil related to this article. Dr. Grodofsky is an equity holder/advisor in Thermaquil relevant to this paper. Ms. Scherer has no conflict of interest to disclose for this work. Dr. Schetzner has no conflict of interest to disclose for this work. Dr. Klusek has no conflict of interest to disclose for this work. Dr. Popielarski is founder and employee of Thermaquil relevant to this paper. Dr. Meloni an employee at Thermaquil relevant to the paper. Dr. Falowski is an equity holder/advisor in Thermaquil related to this article and consultant and took part in research for Aurora, Mainstay, Medtronic, Abbott, Vertiflex, Saluda, Nalu, CornerLoc, Biotronik; equity from SurgenTec, SynerFuse, Aurora Spine, Thermaquil, SPR Therapeutics, Saluda, CornerLoc, PainTeq, and Stimgenics; consultant for Abott, Medtronic, Saluda, Vertos, CornerLoc, Mainstay, Relievant, and Avanos outside of the submitted work. Dr. Kim is an equity holder/advisor in Thermaquil related to this article. Dr. Slavin is an equity holder/advisor in Thermaquil relevant to the paper and outside of the submitted work, Dr. Slavin and/or his institution have received consulting income, research and educational grants from Medtronic, Abbott, Boston Scientific, ATI, MSEI, Integer, Nurami, and Neuros; he has minor ownership or ownership options in Neuramodix, Thermaquil, Higgs Boson and Stimwave. Dr. Silberstein is an equity holder/advisor in Thermaquil relevant to the paper.
Figures

References
-
- Martelletti P., Schwedt T.J., Lanteri-Minet M., Quintana R., Carboni V., Diener H.C., Ruiz De La Torre E., Craven A., Rasmussen A.V., Evans S., et al. My Migraine Voice Survey: A Global Study of Disease Burden among Individuals with Migraine for Whom Preventive Treatments Have Failed. J. Headache Pain. 2018;19:115. doi: 10.1186/s10194-018-0946-z. - DOI - PMC - PubMed
-
- Friedman B.W., Solorzano C., Esses D., Xia S., Hochberg M., Dua N., Heins A., Sasso P., Bijur P.E., Lipton R.B., et al. Treating Headache Recurrence After Emergency Department Discharge: A Randomized Controlled Trial of Naproxen Versus Sumatriptan. Ann. Emerg. Med. 2010;56:7–17. doi: 10.1016/j.annemergmed.2010.02.005. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

