In Vitro and In Vivo Evidence towards Fibronectin's Protective Effects against Prion Infection
- PMID: 38139358
- PMCID: PMC10743696
- DOI: 10.3390/ijms242417525
In Vitro and In Vivo Evidence towards Fibronectin's Protective Effects against Prion Infection
Abstract
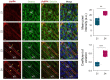
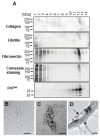
A distinctive signature of the prion diseases is the accumulation of the pathogenic isoform of the prion protein, PrPSc, in the central nervous system of prion-affected humans and animals. PrPSc is also found in peripheral tissues, raising concerns about the potential transmission of pathogenic prions through human food supplies and posing a significant risk to public health. Although muscle tissues are considered to contain levels of low prion infectivity, it has been shown that myotubes in culture efficiently propagate PrPSc. Given the high consumption of muscle tissue, it is important to understand what factors could influence the establishment of a prion infection in muscle tissue. Here we used in vitro myotube cultures, differentiated from the C2C12 myoblast cell line (dC2C12), to identify factors affecting prion replication. A range of experimental conditions revealed that PrPSc is tightly associated with proteins found in the systemic extracellular matrix, mostly fibronectin (FN). The interaction of PrPSc with FN decreased prion infectivity, as determined by standard scrapie cell assay. Interestingly, the prion-resistant reserve cells in dC2C12 cultures displayed a FN-rich extracellular matrix while the prion-susceptible myotubes expressed FN at a low level. In agreement with the in vitro results, immunohistopathological analyses of tissues from sheep infected with natural scrapie demonstrated a prion susceptibility phenotype linked to an extracellular matrix with undetectable levels of FN. Conversely, PrPSc deposits were not observed in tissues expressing FN. These data indicate that extracellular FN may act as a natural barrier against prion replication and that the extracellular matrix composition may be a crucial feature determining prion tropism in different tissues.
Keywords: extracellular matrix (ECM); k fibronectin; muscle; myotube; prion; reserve cells; scrapie; tropism.
Conflict of interest statement
The authors declare no conflict of interest.
Figures








References
-
- Franz M., Eiden M., Balkema-Buschmann A., Greenlee J., Schatzl H., Fast C., Richt J., Hildebrandt J.-P., Groschup M.H. Detection of PrPSc in peripheral tissues of clinically affected cattle after oral challenge with bovine spongiform encephalopathy. J. Gen. Virol. 2012;93:2740–2748. doi: 10.1099/vir.0.044578-0. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

