Impact of SARS-CoV-2 Infection on Humoral and Cellular Immunity in a Cohort of Vaccinated Solid Organ Transplant Recipients
- PMID: 38140248
- PMCID: PMC10747916
- DOI: 10.3390/vaccines11121845
Impact of SARS-CoV-2 Infection on Humoral and Cellular Immunity in a Cohort of Vaccinated Solid Organ Transplant Recipients
Abstract
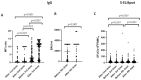
The aim of the present study was to determine humoral and T-cell responses after four doses of mRNA-1273 vaccine in solid organ transplant (SOT) recipients, and to study predictors of immunogenicity, including the role of previous SARS-CoV-2 infection in immunity. Secondarily, safety was also assessed. Liver, heart, and kidney transplant recipients eligible for SARS-CoV-2 vaccination from three different institutions in Barcelona, Spain were included. IgM/IgG antibodies and T cell ELISpot against the S protein four weeks after receiving four consecutive booster doses of the vaccine were analyzed. One hundred and forty-three SOT recipients were included (41% liver, 38% heart, and 21% kidney). The median time from transplantation to vaccination was 6.6 years (SD 7.4). In total, 93% of the patients developed SARS-CoV-2 IgM/IgG antibodies and 94% S-ELISpot positivity. In total, 97% of recipients developed either humoral or cellular response (100% of liver recipients, 95% of heart recipients, and 88% of kidney recipients). Hypogammaglobulinemia was associated with the absence of SARS-CoV-2 IgG/IgM antibodies and S-ELISpot reactivity after vaccination, whereas past symptomatic SARS-CoV-2 infection was associated with SARS-CoV-2 IgG/IgM antibodies and S-ELISpot reactivity. Local and systemic side effects were generally mild or moderate, and no recipients experienced the development of de novo DSA or graft dysfunction following vaccination.
Keywords: COVID-19; cellular; humoral immunity; immunity; solid organ transplant recipients; vaccination.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Velleca A., Shullo M.A., Dhital K., Azeka E., Colvin M., DePasquale E., Farrero M., García-Guereta L., Jamero G., Khush K., et al. The International Society for Heart and Lung Transplantation (ISHLT) guidelines for the care of heart transplant recipients. J. Heart Lung Transpl. 2023;42:e1–e141. doi: 10.1016/j.healun.2022.10.015. - DOI - PubMed
-
- COVID-19 Treatment Guidelines Panel. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health. [(accessed on 7 December 2023)]; Available online: https://www.covid19treatmentguidelines.nih.gov/
LinkOut - more resources
Full Text Sources
Miscellaneous

