In Vitro Pre-Clinical Evaluation of a Gonococcal Trivalent Candidate Vaccine Identified by Transcriptomics
- PMID: 38140249
- PMCID: PMC10747275
- DOI: 10.3390/vaccines11121846
In Vitro Pre-Clinical Evaluation of a Gonococcal Trivalent Candidate Vaccine Identified by Transcriptomics
Abstract
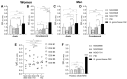
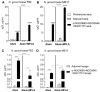
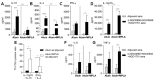
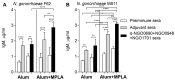
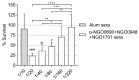
Gonorrhea, a sexually transmitted disease caused by Neisseria gonorrhoeae, poses a significant global public health threat. Infection in women can be asymptomatic and may result in severe reproductive complications. Escalating antibiotic resistance underscores the need for an effective vaccine. Approaches being explored include subunit vaccines and outer membrane vesicles (OMVs), but an ideal candidate remains elusive. Meningococcal OMV-based vaccines have been associated with reduced rates of gonorrhea in retrospective epidemiologic studies, and with accelerated gonococcal clearance in mouse vaginal colonization models. Cross-protection is attributed to shared antigens and possibly cross-reactive, bactericidal antibodies. Using a Candidate Antigen Selection Strategy (CASS) based on the gonococcal transcriptome during human mucosal infection, we identified new potential vaccine targets that, when used to immunize mice, induced the production of antibodies with bactericidal activity against N. gonorrhoeae strains. The current study determined antigen recognition by human sera from N. gonorrhoeae-infected subjects, evaluated their potential as a multi-antigen (combination) vaccine in mice and examined the impact of different adjuvants (Alum or Alum+MPLA) on functional antibody responses to N. gonorrhoeae. Our results indicated that a stronger Th1 immune response component induced by Alum+MPLA led to antibodies with improved bactericidal activity. In conclusion, a combination of CASS-derived antigens may be promising for developing effective gonococcal vaccines.
Keywords: Th1/Th2 responses; adjuvants; bactericidal antibodies; gonorrhea; vaccine.
Conflict of interest statement
S.R. is a cofounder of STIRx, Inc., and holds equity in the company. All other authors declare no conflict of interest.
Figures






References
-
- CDC Sexually Transmitted Disease Surveillance 2021. [(accessed on 23 October 2023)]; Available online: https://www.cdc.gov/std/statistics/2021/overview.htm#Gonorrhea.
-
- Unemo M., Lahra M.M., Escher M., Eremin S., Cole M.J., Galarza P., Ndowa F., Martin I., Dillon J.R., Galas M., et al. WHO global antimicrobial resistance surveillance for Neisseria gonorrhoeae 2017–2018: A retrospective observational study. Lancet Microbe. 2021;2:e627–e636. doi: 10.1016/S2666-5247(21)00171-3. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

