Chromatically Corrected Multicolor Multiphoton Microscopy
- PMID: 38145164
- PMCID: PMC10739991
- DOI: 10.1021/acsphotonics.3c01104
Chromatically Corrected Multicolor Multiphoton Microscopy
Abstract
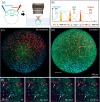
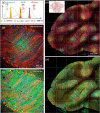
Simultaneous imaging of multiple labels in tissues is key to studying complex biological processes. Although strategies for color multiphoton excitation have been established, chromatic aberration remains a major problem when multiple excitation wavelengths are used in a scanning microscope. Chromatic aberration introduces a spatial shift between the foci of beams of different wavelengths that varies across the field of view, severely degrading the performance of color imaging. In this work, we propose an adaptive correction strategy that solves this problem in two-beam microscopy techniques. Axial chromatic aberration is corrected by a refractive phase mask that introduces pure defocus into one beam, while lateral chromatic aberration is corrected by a piezoelectric mirror that dynamically compensates for lateral shifts during scanning. We show that this light-efficient approach allows seamless chromatic correction over the entire field of view of different multiphoton objectives without compromising spatial and temporal resolution and that the effective area for beam-mixing processes can be increased by more than 1 order of magnitude. We illustrate this approach with simultaneous three-color, two-photon imaging of developing zebrafish embryos and fixed Brainbow mouse brain slices over large areas. These results establish a robust and efficient method for chromatically corrected multiphoton imaging.
© 2023 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
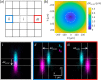
 between the two focused beams across the
imaging field of view. The lateral shifts increase linearly with the
distance from the center of the field of view. White dashed lines
correspond to lateral shifts of 0.80 μm. Microscope objective
used: Zeiss W Plan-Apochromat 20×/1.0 DIC (Zeiss 20×).
between the two focused beams across the
imaging field of view. The lateral shifts increase linearly with the
distance from the center of the field of view. White dashed lines
correspond to lateral shifts of 0.80 μm. Microscope objective
used: Zeiss W Plan-Apochromat 20×/1.0 DIC (Zeiss 20×).
References
LinkOut - more resources
Full Text Sources
