Digital consults to optimize guideline-directed therapy: design of a pragmatic multicenter randomized controlled trial
- PMID: 38146630
- PMCID: PMC10804150
- DOI: 10.1002/ehf2.14634
Digital consults to optimize guideline-directed therapy: design of a pragmatic multicenter randomized controlled trial
Abstract
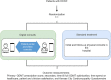
Aims: Many heart failure (HF) patients do not receive optimal guideline-directed medical therapy (GDMT) despite clear benefit on morbidity and mortality outcomes. Digital consults (DCs) have the potential to improve efficiency on GDMT optimization to serve the growing HF population. The investigator-initiated ADMINISTER trial was designed as a pragmatic multicenter randomized controlled open-label trial to evaluate efficacy and safety of DC in patients on HF treatment.
Methods and results: Patients (n = 150) diagnosed with HF with a reduced ejection fraction will be randomized to DC or standard care (1:1). The intervention group receives multifaceted DCs including (i) digital data sharing (e.g. exchange of pharmacotherapy use and home-measured vital signs), (ii) patient education via an e-learning, and (iii) digital guideline recommendations to treating clinicians. The consults are performed remotely unless there is an indication to perform the consult physically. The primary outcome is the GDMT prescription rate score, and secondary outcomes include time till full GDMT optimization, patient and clinician satisfaction, time spent on healthcare, and Kansas City Cardiomyopathy Questionnaire. Results will be reported in accordance to the CONSORT statement.
Conclusions: The ADMINISTER trial will offer the first randomized controlled data on GDMT prescription rates, time till full GDMT optimization, time spent on healthcare, quality of life, and patient and clinician satisfaction of the multifaceted patient- and clinician-targeted DC for GDMT optimization.
Keywords: Clinical trial; Digital health; Guideline adherence; Heart failure.
© 2023 The Authors. ESC Heart Failure published by John Wiley & Sons Ltd on behalf of European Society of Cardiology.
Conflict of interest statement
Dr M.L.H. is supported by the Dutch Heart Foundation (Dr E. Dekker Senior Clinical Scientist Grant 2020T058) and CVON (2020B008 RECONNEXT). Dr M.L.H. has received an investigator‐initiated research grant from Vifor Pharma, an educational grant from Boehringer Ingelheim and Novartis, and speaker/consultancy fees from Abbott, AstraZeneca, Bayer, Boehringer Ingelheim, MSD, Novartis, Sankyo, Daiichi, Quin, and Vifor Pharma.
Figures
References
-
- McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Böhm M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J 2021;42:3599‐3726. - PubMed
-
- McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Böhm M, et al. 2023 Focused update of the 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the task force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J 2023;ehad195. doi:10.1093/eurheartj/ehad195 - DOI - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous