Evidence for distinct mechanisms of immune suppression in EBV-positive and EBV-negative Hodgkin lymphoma
- PMID: 38148013
- PMCID: PMC10861371
- DOI: 10.3960/jslrt.23037
Evidence for distinct mechanisms of immune suppression in EBV-positive and EBV-negative Hodgkin lymphoma
Abstract
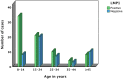
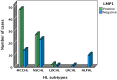
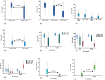
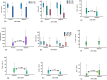
Epstein Barr Virus (EBV) has been recognized for its ability to transform B lymphocytes and for its association with different types of cancers including Hodgkin lymphoma. In addition, EBV may also modulate the microenvironment of HL. In this study, we aimed to investigate the prevalence of EBV among HL cases in Ethiopia and to assess the tissue cellular composition of EBV-related and EBV-unrelated cases. We constructed a tissue microarray (TMA) of 126 consecutive cases of classical HL (CHL) and nodular lymphocyte predominant HL (NLPHL) from a tertiary cancer centre, Tikur Anbessa Hospital, Addis Ababa, Ethiopia, and evaluated a panel of immunohistochemical markers. The quantification of immune cells was performed using HALO 2.3, a platform for image analysis from Indica Lab Inc. A total of 77/126 (61.1%) of HL cases expressed LMP1/EBER. Infiltration of CD8+, T-bet+ and FoxP3+ cells was higher in the microenvironment of EBV-related CHL, with P values of <0.001, <0.001 and <0.016, respectively. In contrast, the expression of PD1 was higher in the microenvironment of EBV-unrelated CHL cases (P < 0.001). Unlike in Western countries, the majority of HL cases in Ethiopia were associated with EBV. As FoxP3+ and PD1-expressing cells are thought to participate in down regulation of the immune response by different mechanisms, this finding highlights the previously unrecognized possibility that distinct immunosuppressive mechanisms may be ongoing within EBV positive and negative HL types. This may have important prognostic and therapeutic implications.
Keywords: Epstein Barr Virus; HL microenvironment; immune suppression.
Conflict of interest statement
CONFLICT OF INTEREST
The authors report no conflicts of interest in this work. Except for Mats Jerkeman, who has research support: Abbvie, AstraZeneca, BMS, Roche, Janssen, Gilead, Genmab, Incyte, BeiGene
Figures
Similar articles
-
Classical Hodgkin Lymphoma with Positive Epstein-Barr Virus Status is Associated with More FOXP3 Regulatory T Cells.Med Sci Monit. 2016 Jul 5;22:2340-6. doi: 10.12659/msm.896629. Med Sci Monit. 2016. PMID: 27377121 Free PMC article.
-
The circuitry of the tumor microenvironment in adult and pediatric Hodgkin lymphoma: cellular composition, cytokine profile, EBV, and exosomes.Cancer Rep (Hoboken). 2021 Apr;4(2):e1311. doi: 10.1002/cnr2.1311. Epub 2020 Oct 26. Cancer Rep (Hoboken). 2021. PMID: 33103852 Free PMC article. Review.
-
Tumor microenvironment composition in pediatric classical Hodgkin lymphoma is modulated by age and Epstein-Barr virus infection.Int J Cancer. 2012 Sep 1;131(5):1142-52. doi: 10.1002/ijc.27314. Epub 2011 Dec 21. Int J Cancer. 2012. PMID: 22025264
-
Epstein-Barr virus recruits PDL1-positive cells at the microenvironment in pediatric Hodgkin lymphoma.Cancer Immunol Immunother. 2021 Jun;70(6):1519-1526. doi: 10.1007/s00262-020-02787-2. Epub 2020 Nov 12. Cancer Immunol Immunother. 2021. PMID: 33184699 Free PMC article.
-
Hodgkin lymphoma: a review of pathological features and recent advances in pathogenesis.Pathology. 2020 Jan;52(1):154-165. doi: 10.1016/j.pathol.2019.09.005. Epub 2019 Nov 5. Pathology. 2020. PMID: 31699300 Review.
Cited by
-
Epstein-Barr Infection, Hodgkin's Lymphoma, and the Immune System: Insights into the Molecular Mechanisms Facilitating Immune Evasion.Cancers (Basel). 2025 Apr 28;17(9):1481. doi: 10.3390/cancers17091481. Cancers (Basel). 2025. PMID: 40361408 Free PMC article. Review.
-
Transition From Epstein-Barr Virus (EBV)-Positive Rectal Hodgkin Lymphoma to Diffuse Large B-Cell Lymphoma in the Lung.Cureus. 2024 Jul 20;16(7):e65013. doi: 10.7759/cureus.65013. eCollection 2024 Jul. Cureus. 2024. PMID: 39165470 Free PMC article.
References
-
- Thorley-Lawson DA, Gross A. Persistence of the Epstein-Barr virus and the origins of associated lymphomas. N Engl J Med. 2004; 350: 1328-1337. - PubMed
-
- Fitzmaurice C, Allen C, Barber RM, et al. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 32 cancer groups, 1990 to 2015: a systematic analysis for the global burden of disease study. JAMA Oncol. 2017; 3: 524-548. - PMC - PubMed
-
- Biggar RJ, Henle W, Fleisher G, et al. Primary Epstein-Barr virus infections in African infants. I. Decline of maternal antibodies and time of infection. Int J Cancer. 1978; 22: 239-243. - PubMed
-
- Cohen JI. Epstein-Barr virus infection. N Engl J Med. 2000; 343: 481-492. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

