Effect of Psychological and Medication Therapies for Insomnia on Daytime Functions: A Randomized Clinical Trial
- PMID: 38153735
- PMCID: PMC10755607
- DOI: 10.1001/jamanetworkopen.2023.49638
Effect of Psychological and Medication Therapies for Insomnia on Daytime Functions: A Randomized Clinical Trial
Abstract
Importance: Daytime functional impairments are the primary reasons for patients with insomnia to seek treatment, yet little is known about what the optimal treatment is for improving daytime functions and how best to proceed with treatment for patients whose insomnia has not remitted.
Objectives: To compare the efficacy of behavioral therapy (BT) and zolpidem as initial therapies for improving daytime functions among patients with insomnia and evaluate the added value of a second treatment for patients whose insomnia has not remitted.
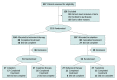
Design, setting, and participants: In this sequential multiple-assignment randomized clinical trial conducted at institutions in Canada and the US, 211 adults with chronic insomnia disorder were enrolled between May 1, 2012, and December 31, 2015, and followed up for 12 months. Statistical analyses were performed on an intention-to-treat basis in April and October 2023.
Interventions: Participants were randomly assigned to either BT or zolpidem as first-stage therapy, and those whose insomnia had not remitted received a second-stage psychological therapy (BT or cognitive therapy) or medication therapy (zolpidem or trazodone).
Main outcomes and measures: Study outcomes were daytime symptoms of insomnia, including mood disturbances, fatigue, functional impairments of insomnia, and scores on the 36-item Short-Form Health Survey (SF-36) physical and mental health components.
Results: Among 211 adults with insomnia (132 women [63%]; mean [SD] age, 45.6 [14.9] years), 104 were allocated to BT and 107 to zolpidem at the first stage. First-stage treatment with BT or zolpidem yielded significant and equivalent benefits for most of the daytime outcomes, including depressive symptoms (Beck Depression Inventory-II mean score change, -3.5 [95% CI, -4.7 to -2.3] vs -4.3 [95% CI, -5.7 to -2.9]), fatigue (Multidimensional Fatigue Inventory mean score change, -4.7 [95% CI, -7.3 to -2.2] vs -5.2 [95% CI, -7.9 to -2.5]), functional impairments (Work and Social Adjustment Scale mean score change, -5.0 [95% CI, -6.7 to -3.3] vs -5.1 [95% CI, -7.2 to -2.9]), and mental health (SF-36 mental health subscale mean score change, 3.5 [95% CI, 1.9-5.1] vs 2.5 [95% CI, 0.4-4.5]), while BT produced larger improvements for anxiety symptoms relative to zolpidem (State-Trait Anxiety Inventory mean score change, -4.1 [95% CI, -5.8 to -2.4] vs -1.2 [95% CI, -3.0 to 0.5]; P = .02; Cohen d = 0.55). Second-stage therapy produced additional improvements for the 2 conditions starting with zolpidem at posttreatment in fatigue (Multidimensional Fatigue Inventory mean score change: zolpidem plus BT, -3.8 [95% CI, -7.1 to -0.4]; zolpidem plus trazodone, -3.7 [95% CI, -6.3 to -1.1]), functional impairments (Work and Social Adjustment Scale mean score change: zolpidem plus BT, -3.7 [95% CI, -6.4 to -1.0]; zolpidem plus trazodone, -3.3 [95% CI, -5.9 to -0.7]) and mental health (SF-36 mental health subscale mean score change: zolpidem plus BT, 5.3 [95% CI, 2.7-7.9]; zolpidem plus trazodone, 2.0 [95% CI, 0.1-4.0]). Treatment benefits achieved at posttreatment were well maintained throughout the 12-month follow-up, and additional improvements were noted for patients receiving the BT treatment sequences.
Conclusions and relevance: In this randomized clinical trial of adults with insomnia disorder, BT and zolpidem produced improvements for various daytime symptoms of insomnia that were no different between treatments. Adding a second treatment offered an added value with further improvements of daytime functions.
Trial registration: ClinicalTrials.gov Identifier: NCT01651442.
Conflict of interest statement
Figures
References
-
- American Psychiatric Association . Diagnostic and Statistical Manual of Mental Disorders. 5th ed. American Psychiatric Association; 2013.
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous