Characteristics of gut microbiota in captive Asian elephants (Elephas maximus) from infant to elderly
- PMID: 38155244
- PMCID: PMC10754835
- DOI: 10.1038/s41598-023-50429-1
Characteristics of gut microbiota in captive Asian elephants (Elephas maximus) from infant to elderly
Abstract
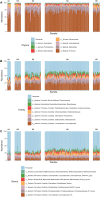
Gut microbiota play an important role in the health and disease of Asian elephants, however, its characteristics at each stage of life have not been thoroughly investigated in maintaining and regulating health of elephants. This study, therefore, aimed to characterize the profiles of the gut microbiota of captive Asian elephants from infants to the elderly. Gut microbiota were identified by 16S rRNA sequencing from the feces of captive Asian elephants with varying age groups, including infant calves, suckling calves, weaned calves, subadult and adult elephants, and geriatric elephants. The diversity of the gut microbiota was lowest in infants, stable during adulthood, and slightly decreased in the geriatric period. The gut microbiota of the infant elephants was dominated by milk-fermenting taxa including genus Bifidobacterium of family Bifidobacteriaceae together with genus Akkermansia. The fiber-fermenting taxa such as Lachnospiraceae_NK3A20_group were found to be increased in suckling elephants in differential abundance analysis by Analysis of Compositions of Microbiomes with Bias Correction (ANCOM-BC). The gut microbiota profiles after weaning until the adult period has been uniform as indicated by no significant differences in beta diversity between groups. However, the composition of the gut microbiota was found to change again in geriatric elephants. Understanding of the composition of the gut microbiota of captive Asian elephants at various life stages could be beneficial for promoting good health throughout their lifespan, as well as ensuring the welfare of captive elephants.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
Characteristics of gut microbiota profiles in Asian elephants (Elephas maximus) with gastrointestinal disorders.Sci Rep. 2025 Jan 8;15(1):1327. doi: 10.1038/s41598-025-85495-0. Sci Rep. 2025. PMID: 39779898 Free PMC article.
-
Mechanism of inulin in colic and gut microbiota of captive Asian elephant.Microbiome. 2023 Jul 6;11(1):148. doi: 10.1186/s40168-023-01581-3. Microbiome. 2023. PMID: 37408039 Free PMC article.
-
Clinical health issues, reproductive hormones, and metabolic hormones associated with gut microbiome structure in African and Asian elephants.Anim Microbiome. 2021 Dec 20;3(1):85. doi: 10.1186/s42523-021-00146-9. Anim Microbiome. 2021. PMID: 34930501 Free PMC article.
-
Analysis of infant microbiota composition and the relationship with breast milk components in the Asian elephant (Elephas maximus) at the zoo.J Vet Med Sci. 2020 Jul 31;82(7):983-989. doi: 10.1292/jvms.20-0190. Epub 2020 Apr 29. J Vet Med Sci. 2020. PMID: 32350162 Free PMC article.
-
Chemical signals in the reproduction of Asian (Elephas maximus) and African (Loxodonta africana) elephants.Anim Reprod Sci. 1998 Oct;53(1-4):19-34. doi: 10.1016/s0378-4320(98)00124-9. Anim Reprod Sci. 1998. PMID: 9835364 Review.
Cited by
-
Characteristics of gut microbiota profiles in Asian elephants (Elephas maximus) with gastrointestinal disorders.Sci Rep. 2025 Jan 8;15(1):1327. doi: 10.1038/s41598-025-85495-0. Sci Rep. 2025. PMID: 39779898 Free PMC article.
-
What Can the Gut Microbiota of Animals Teach Us about the Relationship between Nutrition and Burden of Lifestyle Diseases?Nutrients. 2024 Jun 6;16(11):1789. doi: 10.3390/nu16111789. Nutrients. 2024. PMID: 38892721 Free PMC article. Review.
-
Comparative Analysis of Gut Microbiome Community Structures in Different Populations of Asian Elephants in China and Their Correlation with Diet.Genes (Basel). 2025 Apr 25;16(5):483. doi: 10.3390/genes16050483. Genes (Basel). 2025. PMID: 40428305 Free PMC article.
References
-
- Greene W, Dierenfeld ES, Mikota S. A review of Asian and African elephant gastrointestinal anatomy, physiology, and pharmacology: Elephant gastrointestinal anatomy, physiology, and pharmacology. J. Zoo Aquar. Res. 2019;7:1–14. doi: 10.19227/jzar.v7i1.329. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

